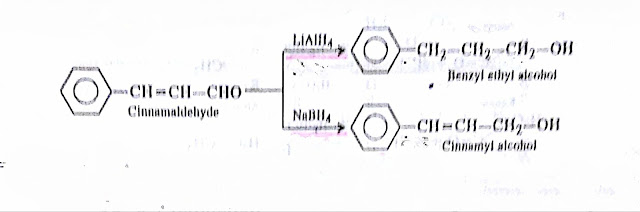
(1) NaBH4 is a milder and selective reducing agent than LiAIH4. It can reduce aldehydes, ketones and acid chlorides without attacking other reducible groups such as esters epoxides, catboxylic acids, esters, amides olefIns etc.
(2) NaBH4 is also used in the second step of the Oxymercuration Demurcuration addition reaction to replace mercury (Hg)(Demurcuration) with H
NaBH4 can reduces:
(1) Aldehydes into primary alcohols.
(2) Ketones into secondary alcohols.
(3) Acid chlorides into primaryalcohols.
NaBH4 can't reduce:
Acids, Amides, Esters, anhydrides, Nitriles, Isonitriles, alkenes, alkynes etc.