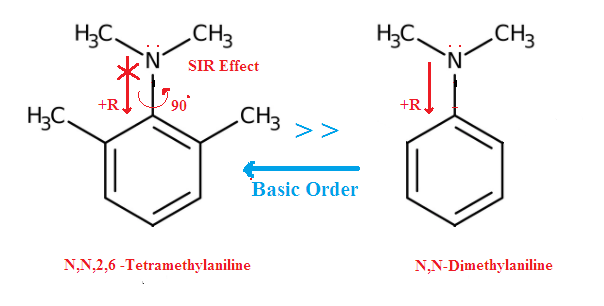
Tertiary amines are more basic than secondary amines, which are more basic than primary because in the gas phase, their is less stearic effect as well as no solvation effect so amines exhibit their basicities due to the electron-releasing effects of the organic substituents only , Thus 3° amines, has three ERG groups so it becomes more basic and ammonia is least basic than amines because their is no ERG .
Related Questions:
- What is relative basic strength order 1° amines , 2°amines and 3° amines ?
- Explain:Arrange the following in the increasing order of their acid strength : Methyl amine, Dimethyl amine, Aniline, N-methyl aniline (I.I.T, 1988).
- What is Carbylamine test or Isocyanide test ?
- What is Hinsberg's reagent and where Hinsberg's test use?
- Can we prepare aniline by Gabriel – phthalimide reaction?
- N-Ethyl-N-methyl propanamine does not show optical activity why?
- What is relative basic strength order 1° amines , 2°amines and 3° amines ? Explain:
- Amines are more basic than ammonia why?
- Give an explanation for the fact that Guanidine NH=C(CH3)2 is a stronger base than most of amines?
- Arrange in correct order of basic Character of aniline, pyrrol, pyridine and piperidine?
- What is correct basicity order of pyridine, pyridazine, pyrimidine and pyrazine ?
- Give an explanation for the fact that Guanidine NH=C(CH3)2 is a stronger base than most of amines?
- Arrange in correct order of basic Character of aniline, pyrrol, pyridine and piperidine?
- What is correct basicity order of pyridine, pyridazine, pyrimidine and pyrazine ?
- Why pyridine is more basic than Pyrrole?
- Why pyrimidine is less basic than pyridine?
- Imidazole is more basic than pyridine? Why?
- Biological Important of Imidazole and structure:
- Pyridine is almost 1 million times less basic than piperidine? Why?
- Cyclohexylamine amine is the stronger base than Aniline? Why?
- Tetrahydroquinoline amine is the stronger base than Tetrahydroisoquinoline? Why?