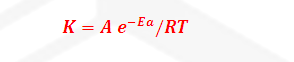Search This Blog
Sunday, May 26, 2024
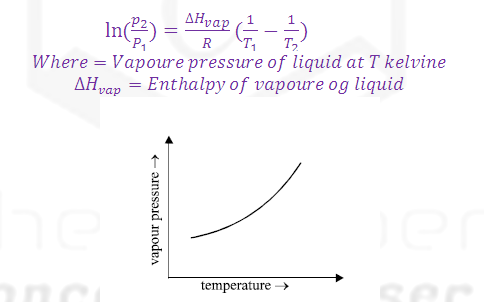
Derivation of Van't Hoff equation.
Tuesday, September 5, 2023
A certain Zero order reaction has rate constant K is 0.025 MS^-1 for disappearance of A . What will be the concentration of A after 15 sec if initial concentration is A is 0.5M.
The rate constant for zero order reaction is 2× 10^-2 Mole L^-1 sec-1. If the concentration of reactant after 25 sec is 0.5M ,the initial concentration must have been ?
Consider a general reaction A--->B follow zero order kinetics.if concentration of A is reduces from 0.1 M to 0.05 M in 10 second then calculate time taken when concentration reduce to 0.025 M.
Thursday, November 25, 2021
A metal slowly forms an oxide film which completely protects the metal when the film thickness is 3.956 thousands of an inch. If the film thickness is 1.281 thousands. in 6 weeks, how much longer will it be before it is 2.481 thou? The rate of film formation follows first order kinetics.
Tuesday, November 23, 2021
Class Claperon equation:
Monday, May 10, 2021
In Arrhenius equation for a certain reaction, the value of A and Ea (activation energy) are 4x 10^13 sec^-1 and 98.6 kJ mole^-1 respectively. At what temperature, the reaction will have specific rate constant 1.1x 10^-3 sec^-1.
Arrhenius equation:
The temperature
dependence of rate of a chemical reaction can be accurately explained by
Arrhenius equation. It was first proposed by Dutch chemist J.H.
Vant’s Hoff but Swedish chemist Arrhenius
provides its physical justification and interpretation.
Where
K=
Rate constant
A= Arrhenius constant or frequency
factor or pre exponential factor
R= Universal gas constant =25/3 joule
per mole per second
Ea= Activation
Energy
T= temperature
-Ea/RT= Boltzmann factor or fraction of molecule having equal or greater
than Activation energy or fraction of molecule that have
kinetic energy greater than activation energy.
Related Questions:
(1) What is Arrhenius equation ?
(2) what happen when activation energy of a chemical reaction becomes equal to RT?
(3) What is the activation energy and how to different from threshold energy ?
(4) In Arrhenius equation for a certain reaction, the value of A and Ea (activation energy) are 4 × 10^13 sec–1 and 98.6 kJ mol–1 respectively. At what temperature, the reaction will have specific rate constant 1.1 × 10^–3 sec–1?
(5) The energy of activation for a reaction is 100 kJ mol–1. Presence of a catalyst lowers the energy of activation by 75%.What will be effect on rate of reaction at 20ºC, other things being equal?
(6) A drug becomes ineffective after 30 % decomposition. The original concentration of a sample was 5mg/mL which becomes 4.2 mg/mL during 20 months. Assuming the decomposition of first order , calculate the expiry time of the drug in months. What is the half life of the product?
(7) A first order reaction is 20 % completed in 10 minutes. Calculate the time taken for the reaction to go to 80 % completion.
(8) The rate of a reaction triple when temperature changes from 20”C to 50”C. Calculate energy of activation for the reaction (R = 8.314 JK^1 mol^1).
Thursday, October 1, 2020
The energy of activation for a reaction is 100 kJ mol^-1. Presence of a catalyst lowers the energy of activation by 75%. What will be effect on rate of reaction at 20”C, other things being equal ?
Related Questions:
What is Arrhenius equation ?
what happen when activation energy of a chemical reaction becomes equal to RT?
What is the activation energy and how to different from threshold energy ?
In Arrhenius equation for a certain reaction, the value of A and Ea (activation energy) are 4 × 10^13 sec–1 and 98.6 kJ mol–1 respectively. At what temperature, the reaction will have specific rate constant 1.1 × 10^–3 sec–1?
The energy of activation for a reaction is 100 kJ mol–1. Presence of a catalyst lowers the energy of activation by 75%.What will be effect on rate of reaction at 20ºC, other things being equal?
A drug becomes ineffective after 30 % decomposition. The original concentration of a sample was 5mg/mL which becomes 4.2 mg/mL during 20 months. Assuming the decomposition of first order , calculate the expiry time of the drug in months. What is the half life of the product?
A first order reaction is 20 % completed in 10 minutes. Calculate the time taken for the reaction to go to 80 % completion.
The rate of a reaction triple when temperature changes from 20”C to 50”C. Calculate energy of activation for the reaction (R = 8.314 JK^1 mol^1).
In Arrhenius equation for a certain reaction, the value of A and Ea (activation energy) are 4 x10^13 sec^1 and 98.6 kJ mol1 respectively. At what temperature, the reaction will have specific rate constant 1.1x10^-3 sec^-1 ?
At 407 K the rate constant of a chemical reaction is 9.5 x10^-5s^-1 and at 420 K, the rate constant is 1.9 x10^-4s^-1. Calculate the frequency factor of the reaction.
At 407 K the rate constant of a chemical reaction is 9.5 x10^-5s^-1 and at 420 K, the rate constant is 1.9 x10^-4s^-1. Calculate the frequency factor of the reaction.
Related Questions:
What is Arrhenius equation ?
what happen when activation energy of a chemical reaction becomes equal to RT?
What is the activation energy and how to different from threshold energy ?
In Arrhenius equation for a certain reaction, the value of A and Ea (activation energy) are 4 × 10^13 sec–1 and 98.6 kJ mol–1 respectively. At what temperature, the reaction will have specific rate constant 1.1 × 10^–3 sec–1?
The energy of activation for a reaction is 100 kJ mol–1. Presence of a catalyst lowers the energy of activation by 75%.What will be effect on rate of reaction at 20ºC, other things being equal?
A drug becomes ineffective after 30 % decomposition. The original concentration of a sample was 5mg/mL which becomes 4.2 mg/mL during 20 months. Assuming the decomposition of first order , calculate the expiry time of the drug in months. What is the half life of the product?
A first order reaction is 20 % completed in 10 minutes. Calculate the time taken for the reaction to go to 80 % completion.
The rate of a reaction triple when temperature changes from 20”C to 50”C. Calculate energy of activation for the reaction (R = 8.314 JK^1 mol^1).
In Arrhenius equation for a certain reaction, the value of A and Ea (activation energy) are 4 x10^13 sec^1 and 98.6 kJ mol1 respectively. At what temperature, the reaction will have specific rate constant 1.1x10^-3 sec^-1 ?
In Arrhenius equation for a certain reaction, the value of A and Ea (activation energy) are 4 x10^13 sec^1 and 98.6 kJ mol1 respectively. At what temperature, the reaction will have specific rate constant 1.1x10^-3 sec^-1 ?
Related Questions:
What is Arrhenius equation ?
what happen when activation energy of a chemical reaction becomes equal to RT?
What is the activation energy and how to different from threshold energy ?
In Arrhenius equation for a certain reaction, the value of A and Ea (activation energy) are 4 × 10^13 sec–1 and 98.6 kJ mol–1 respectively. At what temperature, the reaction will have specific rate constant 1.1 × 10^–3 sec–1?
The energy of activation for a reaction is 100 kJ mol–1. Presence of a catalyst lowers the energy of activation by 75%.What will be effect on rate of reaction at 20ºC, other things being equal?
A drug becomes ineffective after 30 % decomposition. The original concentration of a sample was 5mg/mL which becomes 4.2 mg/mL during 20 months. Assuming the decomposition of first order , calculate the expiry time of the drug in months. What is the half life of the product?
A first order reaction is 20 % completed in 10 minutes. Calculate the time taken for the reaction to go to 80 % completion.
The rate of a reaction triple when temperature changes from 20”C to 50”C. Calculate energy of activation for the reaction (R = 8.314 JK^1 mol^1).
The rate of a reaction triple when temperature changes from 20”C to 50”C. Calculate energy of activation for the reaction (R = 8.314 JK^-1 mol^-1).
Related Questions:
What is Arrhenius equation ?
what happen when activation energy of a chemical reaction becomes equal to RT?
What is the activation energy and how to different from threshold energy ?
In Arrhenius equation for a certain reaction, the value of A and Ea (activation energy) are 4 × 10^13 sec–1 and 98.6 kJ mol–1 respectively. At what temperature, the reaction will have specific rate constant 1.1 × 10^–3 sec–1?
The energy of activation for a reaction is 100 kJ mol–1. Presence of a catalyst lowers the energy of activation by 75%.What will be effect on rate of reaction at 20ºC, other things being equal?
A drug becomes ineffective after 30 % decomposition. The original concentration of a sample was 5mg/mL which becomes 4.2 mg/mL during 20 months. Assuming the decomposition of first order , calculate the expiry time of the drug in months. What is the half life of the product?
A first order reaction is 20 % completed in 10 minutes. Calculate the time taken for the reaction to go to 80 % completion.
A first order reaction is 20 % completed in 10 minutes. Calculate the time taken for the reaction to go to 80 % completion.
Related Questions:
What is Arrhenius equation ?
what happen when activation energy of a chemical reaction becomes equal to RT?
What is the activation energy and how to different from threshold energy ?
In Arrhenius equation for a certain reaction, the value of A and Ea (activation energy) are 4 × 10^13 sec–1 and 98.6 kJ mol–1 respectively. At what temperature, the reaction will have specific rate constant 1.1 × 10^–3 sec–1?
The energy of activation for a reaction is 100 kJ mol–1. Presence of a catalyst lowers the energy of activation by 75%.What will be effect on rate of reaction at 20ºC, other things being equal?
A drug becomes ineffective after 30 % decomposition. The original concentration of a sample was 5mg/mL which becomes 4.2 mg/mL during 20 months. Assuming the decomposition of first order , calculate the expiry time of the drug in months. What is the half life of the product?
A drug becomes ineffective after 30 % decomposition. The original concentration of a sample was 5mg/mL which becomes 4.2 mg/mL during 20 months. Assuming the decomposition of first order , calculate the expiry time of the drug in months. What is the half life of the product?

Related Questions:
What is Arrhenius equation ?
what happen when activation energy of a chemical reaction becomes equal to RT?
What is the activation energy and how to different from threshold energy ?
In Arrhenius equation for a certain reaction, the value of A and Ea (activation energy) are 4 × 10^13 sec–1 and 98.6 kJ mol–1 respectively. At what temperature, the reaction will have specific rate constant 1.1 × 10^–3 sec–1?
The energy of activation for a reaction is 100 kJ mol–1. Presence of a catalyst lowers the energy of activation by 75%.What will be effect on rate of reaction at 20ºC, other things being equal?
Monday, September 21, 2020
Chemical Kinetics
Tuesday, July 21, 2020
Decomposition of H2O2 follows a first order reaction. In fifty minutes the concentration of H2O2 decreases from 0.5 to 0.125 M in one such decomposition. When the concentration of H2O2 reaches 0.05 M, the rate of formation of O2 will be : [JEE MAINS 2016]
Saturday, June 27, 2020
What is Arrhenius equation ?
The temperature
dependence of rate of a chemical reaction can be accurately explained by
Arrhenius equation. It was first proposed by Dutch chemist J.H.
Vant’s Hoff but Swedish chemist Arrhenius
provides its physical justification and interpretation.
Where
K=
Rate constant
A= Arrhenius constant or frequency
factor or pre exponential factor
R= Universal gas constant =25/3 joule
per mole per second
Ea= Activation
Energy
T= temperature
-Ea/RT= Boltzmann factor or fraction of molecule having equal or greater
than Activation energy or fraction of molecule that have
kinetic energy greater than activation energy.
Related Questions: