The temperature
dependence of rate of a chemical reaction can be accurately explained by
Arrhenius equation. It was first proposed by Dutch chemist J.H.
Vant’s Hoff but Swedish chemist Arrhenius
provides its physical justification and interpretation.
Where
K=
Rate constant
A= Arrhenius constant or frequency
factor or pre exponential factor
R= Universal gas constant =25/3 joule
per mole per second
Ea= Activation
Energy
T= temperature
-Ea/RT= Boltzmann factor or fraction of molecule having equal or greater
than Activation energy or fraction of molecule that have
kinetic energy greater than activation energy.
Related Questions:

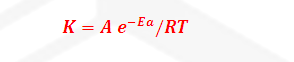
nice
ReplyDelete