(1) Oleum can be
represented by the formula ySO3.H2O where
y is the total molar sulphur trioxide content .the value of y can be varied to
different Oleum sample.
(2) Oleum also be expressed as H2SO4.xSO3 where x is molar free suphur trioxide.
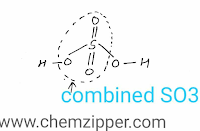
(3) Oleum is the solution of of sulphur trioxide in sulphuric acid , it is also known as fuming sulphuric acid or Pyrosulphuric acid (H2S2O7=H2SO4+SO3).
(4) Oleum sample contain two type of SO3.
(i) Free SO3:- It is that SO3 which combined with water water to give H2SO4 .
PERCENTAGE(%) LABELLING OF OLEUM
:
OLEUM concentration represented by
(100+X) like 104.5%, 109%, 113.4% ,118% etc.
" (100+X)% labelling of Oleum defined as a number which is greater than 100, it mean (X ) is the amount of water in gm required to destroyed free SO3 completely, present in 100 gm of oleum sample"
For example: 109% oleum sample means that 9 gm water required to destroyed the free SO3 completely , present in 100 gm of Oleum Sulphate sample.
CALCULATIONS OF FREE SO3 IN (X%) OLEUM SAMPLE :
Given - X % labelled 100 gm Oleum, it means (X-100) gm water required to destroyed all free SO3 present in 100 gm Oleum.
WH2O= (X-100) gm
BY STOICHIOMETRIC CALCULATION:
Weight of SO3 = nSO3 X
Molecular wt SO3
Weight of H2SO4 = nH2SO4 X Molecular wt of H2SO4
Thus % (Free) SO3 present in 100 gm 109% labelled Oleum Sample is
Calculation of maximum % labelling of
Oleum sample:
NOTE-For maximum possible labbelling amount
of SO3 is 100 gm, It means 100 gm Oleum sample contains 100 gm SO3 only and zero( 0 )gm H2SO4
,hence maximum labbelling possible is 122.5 %.
EXAMPLE (1):
Calculate the % of free SO3 in an Oleum sample that is labelled as
118 % ?
SOLUTION:
EXAMPLE (2):
If the percentage free SO3 in an Oleum sample is 20% then label the
sample of Oleum in term of percentage H2SO4.?
SOLUTION:
EXAMPLE(3):
Two sample of Oleum are labelled as 109% and 115%,what is the difference
between weight of free SO3 in these samples ?.
SOLUTION: Given % labelling (X)=109% and 115% ,find difference between weight of free SO3
in these samples ?.
Difference between weight of free SO3
in these samples are= 66.66-44= 26.67 gm
EXAMPLE(4):
What is the %SO3 in Oleum sample that is labelled as 104.5% H2SO4
?.
EXAMPLE(5):
9 gm water is added into Oleum sample labelled as 112% H2SO4 then the amount of free SO3
remaining in the solution is ? (STP=1atm and 273K).
SOLUTION: Initial free moles of SO3=
Moles of
water that combined with free moles of SO3 =9/18=1/2
moles
Moles of free
SO3 left 2/3-1/2=1/6 moles
Volume
of free SO3 at STP=1/6X22.4=3.73 L
EXAMPLE(6):
Find out the % labelling of oleum
Sulphate in which mole fraction of SO3 is 0.2 ?.
EXAMPLE (7):
Find out the % labelling of new oleum sample obtained by mixing of 4.5 gm of
water in 100 gm of 109% labelled oleum sample ?.
SOLUTION: Wt of SO3 in Original Oleum
The amount of free SO3 destroyed by 4.5 gm
water is added
The amount of free SO3 destroyed by 4.5 gm
water is =
x80=20 gm
Wt of left SO3 =40-20=20 gm
Given Wt of (Free) SO3= 20 % Find % labelling (X)
% labeling(X) =
104.5 %
EXAMPLE(8):
100 gm of 120% labelled Oleum is diluted
with 15 gm of water. determined the new % labelling of Oleum ?.
SOLUTION: : Wt of SO3 in Original Oleum
The amount of free SO3 destroyed by 15 gm water is added
% labelling(X) = 105 %
The amount of free SO3 destroyed by 4.5 gm water=15/18 x 80=66.66 gm
Wt of left SO3 =88.88-66.66=22.22 gm
Given Wt of (Free) SO3= 22.22 % Find % labelling (X)
%
labelling(X) = 105 %
EXAMPLE(9):
Calculate amount of total H2SO4 when 100 gm 109% labelled
Oleum sample is completely destroyed by water ?.
SOLUTION: the amount H2SO4
SOLUTION: the amount H2SO4
Originally 109% 100 gm Oleum sample contains
40 gm free SO3 and 60 gram H2SO4
Hence
total Wt of H2SO4 =60 gm +49 gm =109 gm
EXAMPLE(10):
25 gm of Oleum sample required 2 gm of water ,find out the % labelling of sample
.
SOLUTION: : 25 gm oleum required 2 gm water
1 gm require …………. 2/25 gm
water
100 gm require ……..2/25x100=8
gm
Hence % labelling is 108%
EXAMPLE(11):
A mixture is prepared by mixing of 20 gm SO3 in 30 gm of H2SO4
.
(I)
Find
the mole fraction of SO3 .
(II)
Determine
% labelling of Oleum sample.
SOLUTION:
(i) Total wt of Oleum is 20 gm SO3+
30 gm H2SO4
(ii)
Given Wt of (Free) SO3= 40 % Find % labelling
(X)
% labelling(X) = 109%
EXAMPLE(12):
What volume of 1M NaOH (in ml)will
required to react completely with 100 gm of Oleum which is 109 % labelled ?.
SOLUTION: We know that 109% Oleum sample contains 40 gm SO3 and 60 gm H2SO4
(E wt= SO3=80/2=40
gm and E wt H2SO4 =98/2=49 gm)
At equivalent point
No of equivalent of SO3 + No
of equivalent H2SO4 = no of equivalent of NaOH
EXAMPLE (13)
: 0.5 gm of fume H2SO4 (Oleum ) is diluted with water,
this solution is completely neutralised by 26.7 ml of 0.4 N NaOH. Find the
percentage free SO3 in sample solution.?
SOLUTION: Given total wt of Oleum sample is 0.5 gm, let x gm SO3 and (0.5-x) H2SO4
(E wt=
SO3=80/2=40 gm and E wt H2SO4=98/2=49
gm)
At equivalent
point
No of equivalent of SO3 + No
of equivalent H2SO4 = no of equivalent of NaOH
EXAMPLE (14):
A mixture of H2CO3 liquid and CO2 gas is labelled
as Oleum sample . 50 gm such mixture contains 22% CO2 , find out the % labelling of such mixture.
SOLUTION:
Given 22% CO2
% labeling(X) of CO2 = 109%
EXAMPLE
(15): Calculate how much H2SO4 will be obtained from 400 gm of
Oleum sample having labelling 104.5%?
SOLUTION: 104.5 %
labelled means 100 Oleum sample required 4.5 gm water to completely destroyed
free SO3 present in 100 gm sample
100
gm Oleum sample require 4.5 gm water to destroyed all free SO3
Weight
of SO3 in destroyed by 18 gm water is =18/18x80= 80 gm
Weight of H2SO4
= 400-80= 320 gm present in 400 gm Oleum sample
Weight of H2SO4 newly formed is =18/18x98= 98 gm
Total Weight H2SO4 =320+98=418 gm



























Hats off to your presence of mind..I really enjoyed reading your blog. I really appreciate your information which you shared with us.
ReplyDeleteBest CRM System
super very well explained and easily understood .
ReplyDeleteI am not getting how you got weight of so3 and h2sio4 as((x-100)/18)*(molecular weight)
ReplyDeleteFantastic post as well as great guidance! This article is very useful and helpful for us. Thanks for spreading valuable info.
ReplyDeleteThis is really perfect.
ReplyDeleteKinemaster mod apk
Mod APK
jt express
This is really amazing post
ReplyDelete