SIR stand for Steric Inhibition in Resonance, means as per name steric means size, inhibition means some kind of hindrance,
little hurdle, and resonance means delocalisation of conjugate
points.
We know that planarity is the main
condition for resonance that means resonance can occur only when all the atom
involved in resonance lie in the same plane or nearly in the same plane. Any change
in structure which destroys planarity of molecule will restrict or inhibit
resonance, this phenomenon is known as steric inhibition of resonance.
SIR effect finds immense use in
explaining Stability of carbocation, acidity and basicity of organic compounds:
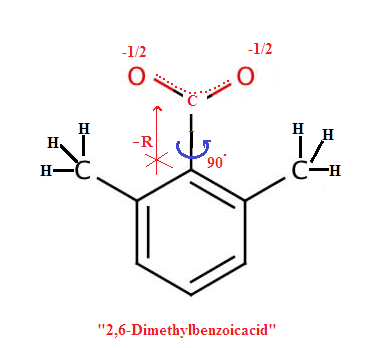
For example(1): Ortho substituted Benzoic acid is more acidic
then meta or para substituted benzoic acid irrespective of nature of group
(electron donating group or electron withdrawing group)
When a group present in the ortho
position with respect to carboxylic group creates steric strain resulting in
rotation of the carboxylic group and shifting it out of plane of the benzene
ring as a result the carboxylic group can no longer participate in ring resonance
and thereby the acidity increases as delocalization of negative charge equally on
the both oxygen atom of conjugate base (
carboxylate ion) of the benzoic acid , hence carboxylate ion more stablised. This
is also called Ortho effect.
Note:
However for groups like -NH2 or -OH does not experience SIR
effect due to small size and –CN is linear group so SIR effect not applicable.
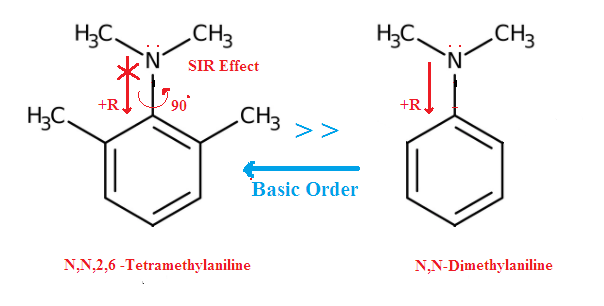
For example (2): N,N,2,6-Tetramethyl aniline is more basic than N,N-Dimethyl aniline. In cas of N,N-Dimethyl
aniline its dialkyl derivatives the NMe2 group is coplanar with
benzene ring. So p-electron (lone
pair) on N atom and pi orbitals are remain in the same plane. For this the
p-electron on N-atom can delocalized via pi orbital in the benzene ring and its
result the electron availability in para position is high and less available at
nitrogen atom and hence less basic.
On the other hand , in case of N,N,2,6 tetramethyl aniline having two bulky methyl
group in ortho position of the benzene ring , the NMe2 group can not
remain in the same plane. That is why the p-electron on N-atom cannot
delocalized through pi orbital in para position. (due to steric inhibition of
resonance) It is localized at nitrogen
atom Thus 2,6-Dimethyl aniline derivative is more basic.

which is more basic N,N dimethylaniline or 2,6 ditertbutylpyridine
ReplyDeletePlz give more explanation and info
ReplyDeleteBht axha 👍
ReplyDeletejee nikla
Delete