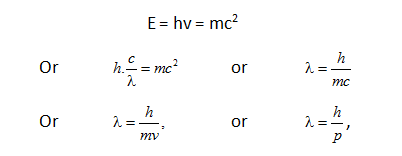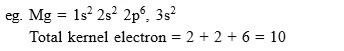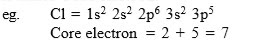Search This Blog
Monday, April 21, 2025
Sunday, August 20, 2023
Saturday, July 3, 2021
What is the "de Broglie equation" or How can derived "de Broglie equation" or What is the "Wave –Particle Duality" ?
Wave –Particle Duality, Dual Nature of Electron:
Bohr’s and
Sommerfield proposed that angular momentum in closed orbit is “Quantized” but
they could not derived mathematically.
Derivation of
this fact was made by Loius de Broglie in 1924 assuming the dual nature of electron
i.e. particle as well as wave nature of electron. This proposal gave birth of a
new theory known as “ Wave Mechanical theory of matter”
Loius de
Broglie was suggested that a particle in motion also behaves like a wave. The wave
length associated with the moving particle is given by the following equation,
Where
h =
Planck’s constant,
m =
mass of the moving particle,
v = velocity
of the particle.
This above equation
(1) is known as de Broglie equation.
De Broglie equation was derived on the basis of Einstein’s equation, E = mc2 and Planck’s equation
E = hv. From both of these relation,
Where p is the momentum of the moving particle.
(1) The wave length associated with a particle in motion is inversely
proportional to its momentum.
(2) As the mass of the moving particle increases, the momentum also
increases; the wave length of the matter wave (associated
with the matter in motion) decreases.
(3) For the particles of finite size, i.e., having appreciable mass, the
momentum is very high, ![]() is very small and it
can be said that macroscopic bodies in motion do not possess matter waves.
is very small and it
can be said that macroscopic bodies in motion do not possess matter waves.
(4) When different particles move with the same velocity, the wave length
of the matter wave is inversely proportional to the mass of the particle.
(1) Show that circumference of the Bohr orbit for hydrogen atom is an integral multiple of the de Broglie wavelength associated with the electron revolving about that orbit.
Solved Questions:
(1) Calculate the energy per mole of photon of electromagnetic radiations of wavelength 4000Å.
What is the de Broglie wave length associated with a proton moving with 25% of the velocity of light?
(1) Show that circumference of the Bohr orbit for hydrogen atom is an integral multiple of the de Broglie wavelength associated with the electron revolving about that orbit.
Solved Questions:
(1) Calculate the energy per mole of photon of electromagnetic radiations of wavelength 4000Å.
The de Broglie wave length of an alpha particle is 6.28 × 10^–15m. Calculate the velocity of the alpha particle (mp = 1.672 × 10^–27kg, mn = 1.675 × 10^–27 kg).
(1) Show that circumference of the Bohr orbit for hydrogen atom is an integral multiple of the de Broglie wavelength associated with the electron revolving about that orbit.
Solved Questions:
(1) Calculate the energy per mole of photon of electromagnetic radiations of wavelength 4000Å.
Calculate the speed and de Broglie wavelength of an electron that has been accelerated by a potential difference of 500 V.
(1) Show that circumference of the Bohr orbit for hydrogen atom is an integral multiple of the de Broglie wavelength associated with the electron revolving about that orbit.
Solved Questions:
(1) Calculate the energy per mole of photon of electromagnetic radiations of wavelength 4000Å.
Practice Questions:
1. A photon of light of wavelength 6000 Å has energy E. Calculate the wavelength of photon of a light which has energy/photon 4E.
2. Calculate the wavelength, wave number and frequency of photon having its energy 5 electron volt.
3. Iodine molecule dissociates into atoms after absorbing light of 4500Å. If one quantum of radiation is absorbed by each molecule, calculate the kinetic energy of iodine atoms. (Bond energy of I2 = 240 kJ mol–1)
Answer Keys:
1. Lemda-2= 1500 Å
2. 2.475 × 10–5 cm, 4.04 × 104 cm, 1.212 × 1015 Hz
3. 2.16 × 10–20 J
Why does a moving cricket ball not show wave like nature?
According to
this equation wave length decreases if m
and u increases. Thus, high mass
object like a cricket ball in flight would have very small wavelength and thus,
do not show any appreciable wave nature.
Related Questions:
What is the difference between 2p and 3p orbitals?
Why are 2p orbitals higher in energy than 2s?
Does 2s and 2p have the same energy?
Calculate radial and angular node for 2p-orbitals.
What are the difference between "Nodal surface" and "Nodal plane"?
What are Zeeman and Stark effects? Were they explained by the Bohr’s theory?
Which of the Hydrogen spectrum series found in visible range of spectrum?
What are difference between "Isoelectronic" and "Isosters" species?
What are the difference between "Kernel" and "Core" ?
Show that circumference of the Bohr orbit for hydrogen atom is an integral multiple of the de Broglie wavelength associated with the electron revolving about that orbit.
Related Questions:
(1) The ionization energy of He+ is 19.6 10^-18 J atom^-1. The energy of the first stationary state of Li+2 will be :
(2) Show that the wavelength of a 150 g rubber ball moving with a velocity 50 m sec^-1 is short enough to be observed ..
(3) If an electron is present in n = 6 level. How many spectral lines would be observed in case of H atom ...
(4) Speed of helium atom at 300K is 2.40 x10^2 meter per sec. What is its wave length. (mass number of helium is 4)..
(5) K.E. of the electron is 4.55 x 10^-25 J. Its de Broglie wave length is ...
(6) The wavelength associated with a golf ball weighing 200g and moving at a speed of 5m/h is of the order.
(7) The ratio of wave length of photon corresponding to the alpha-line of Lyman series in H-atom and Beta-line of Balmer series in He+ is
(8) shortest wavelength of He+ ion in Balmer series is x, then longest wavelength in the Paschene series of Li+2 is
(9) The ratio of difference in wavelengths of 1st and 2nd lines of Lyman series in H like atom to difference in wavelength for 2nd and 3rd lines of same series is:
(10) If H+ (ion) is accelerated to a final velocity of 6.62×10+6 meter per second and error in velocity is 1% then find uncertainty in position is?
(11) What is the relation between Total energy (TE), Kinetic energy (KE) and Potential energy (PE) of Bohr's orbits?
(12) How to calculate energy of Bohr's orbits in term of Rydberg's constant
What is Quantum Theory of Radiation ?
Quantum Theory of Radiation:This theory was proposed by German physicist, Max Planck in 1900, to explain the phenomena of black body radiations. According to this theory;
“Energy is emitted or absorbed by a body not
continuously but discontinuously in the form of small packets or bundles of
energy known as Quanta. The energy of a body can increase or decrease by
1,2,3,4,5,…..n quanta but not by fractions of a quanta."
Thus, the energy of radiation is proportional to its
frequency and inversely proportional to wave length.
Solved Questions:
(1) Calculate the energy per mole of photon of electromagnetic radiations of wavelength 4000Å.
Practice Questions:
1. A photon of light of wavelength 6000 Å has energy E. Calculate the
wavelength of photon of a light which has energy/photon 4E.
2. Calculate the wavelength, wave
number and frequency of photon having its energy 5 electron volt.
3. Iodine
molecule dissociates into atoms after absorbing light of 4500Å. If one quantum
of radiation is absorbed by each molecule, calculate the kinetic energy of iodine
atoms. (Bond energy of I2 = 240 kJ mol–1)
Answer Keys:
1. Lemda-2=
1500 Å
2. 2.475 ×
10–5 cm, 4.04 × 104 cm, 1.212 × 1015 Hz
3. 2.16 × 10–20
J
(1) What is the difference between 2p and 3p orbitals?
(2) Why are 2p orbitals higher in energy than 2s?
(3) Does 2s and 2p have the same energy?
(4) Calculate radial and angular node for 2p-orbitals.
(5) What are the difference between "Nodal surface" and "Nodal plane"?
(6) What are Zeeman and Stark effects? Were they explained by the Bohr’s theory?
(7) Which of the Hydrogen spectrum series found in visible range of spectrum?
(8) What are difference between "Isoelectronic" and "Isosters" species?
(9) What are the difference between "Kernel" and "Core" ?
A bulb emits light of wave length 4500Å. The bulb is rated as 150 watt and 8% of the energy is emitted as light. How many photons are emitted by the bulb per second?
Related Questions:
What is the difference between 2p and 3p orbitals?
Why are 2p orbitals higher in energy than 2s?
Does 2s and 2p have the same energy?
Calculate radial and angular node for 2p-orbitals.
What are the difference between "Nodal surface" and "Nodal plane"?
What are Zeeman and Stark effects? Were they explained by the Bohr’s theory?
Which of the Hydrogen spectrum series found in visible range of spectrum?
What are difference between "Isoelectronic" and "Isosters" species?
What are the difference between "Kernel" and "Core" ?
Suppose 10–17 J of light energy is needed by the interior of the human eye to see an object. How many photons of green light (wavelength =550 nm) are needed to generate this minimum amount of energy?
Related Questions:
(1) The ionization energy of He+ is 19.6 10^-18 J
atom^-1. The energy of the first stationary state of Li+2 will be :
(2) Show that the wavelength of a 150 g rubber ball
moving with a velocity 50 m sec^-1 is short enough to be observed ..
(3) If an electron is present in n = 6 level. How many
spectral lines would be observed in case of H atom ...
(4) Speed of helium atom at 300K is 2.40 x10^2 meter per
sec. What is its wave length. (mass number of helium is 4)..
(5) K.E. of the electron is 4.55 x 10^-25 J. Its de
Broglie wave length is ...
(6) The wavelength associated with a golf ball weighing
200g and moving at a speed of 5m/h is of the order.
(7) The ratio of wave length of photon corresponding to
the alpha-line of Lyman series in H-atom and Beta-line of Balmer series in He+
is
(8) shortest wavelength of He+ ion in Balmer series
is x, then longest wavelength in the Paschene series of Li+2 is
(9) The ratio of difference in wavelengths of 1st and 2nd
lines of Lyman series in H like atom to difference in wavelength for 2nd and
3rd lines of same series is:
(10) If H+ (ion) is accelerated to a final velocity of
6.62×10+6 meter per second and error in velocity is 1% then find uncertainty in
position is?
(11) What is the relation between Total energy (TE),
Kinetic energy (KE) and Potential energy (PE) of Bohr's orbits?
(12) How to calculate energy of Bohr's orbits in term of
Rydberg's constant
Calculate the energy per mole of photon of electromagnetic radiations of wavelength 4000Å.
Related Questions:
(1) On the basis of Heisenbergs uncertainty principle,
show that the electron cannot exist within the nucleus.
(2) Atomic radius is the order of 10^-8 cm. and nuclear
radius is the order of 10^--13 cm. Calculate what fraction of atom is occupied
by nucleus
(3) Electromagnetic radiations of wavelength 242 nm is
just sufficient to ionise sodium atom. Calculate the ionisation energy of
sodium in kJ mol^-1.
(4) The shortest wave length in H spectrum of Lymen
series when Rh = 109678 cm^1 is
(5) An electron beam can undergo diffraction by crystals.
Through what potential should a beam of electrons be accelerated so that its
wavelength becomes equal to 1.54 A
(6) The mass charge ratio for A+ ion is 1.97x10^-7 kg
C^-1. Calculate the mass of A atom.
(7) AIR service on Vividh Bharati is transmitted on 219 m
band. What is its transmission frequency in Hertz?
(8) Calculate the number of photon emitted in 10 hours by
a 60 W sodium lamp (lemda or photon = 5893 A")
(9) Calculate the longest wavelength which can remove the
electron from first Bohrs orbit. Given E1 = 13.6 eV .
(10) Calculate momentum of radiations of wavelength 0.33
nm.
The quantum theory assumes that energy changes are not continuous. Why do not we notice this effect in our everyday activities?
In everyday activities, we deal with macroscopic particles such as our bodies, or cars which gains and loss total amount of energy much larger than a quantum. The gain and loss of the relatively miniscule quantum of energy is unnoticed.
Related Questions:
What is the difference between 2p and 3p orbitals?
Why are 2p orbitals higher in energy than 2s?
Does 2s and 2p have the same energy?
Calculate radial and angular node for 2p-orbitals.
What are the difference between "Nodal surface" and "Nodal plane"?
What are Zeeman and Stark effects? Were they
explained by the Bohr’s theory?
Which of the Hydrogen spectrum series found in
visible range of spectrum?
What are difference between "Isoelectronic"
and "Isosters" species?
What are the difference between "Kernel" and
"Core" ?
Sunday, December 27, 2020
What is the difference between 2p and 3p orbitals?
Importance difference in between 2p and 3p Orbitals is their energy.
The energy of atomic orbitals depends upon principal as well as azimuthal quantum number, which is more common known as (n+l) rule .
According to (n+l) rule the atomic orbital has less (n+l) value also has less energy vice versa.
If the value of (l):is same for two different orbitals then the greater the value of n greater is energy and vice versa.
For 2p => n= 2, l=0,. =>n+l= 2+0=2
For 3p => n= 3, l=0,. =>n+l= 2+0=3
Hence 3p have more energy than 2p orbital
Why are 2p orbitals higher in energy than 2s?
The energy of atomic orbitals depends upon principal as well as azimuthal quantum number, which is more common known as (n+l) rule .
According to (n+l) rule the atomic orbital has less (n+l) value also has less energy vice versa.
For 2S => n= 2, l=0,. =>n+l= 2+0=2
For 2p => n= 2, l=2,. =>n+l= 2+1=3
Hence 2p have more energy than 2s orbital