Search This Blog
Thursday, April 14, 2022
Ellingham Diagram and it's features:
Hall Heroult process : Electrolytic refining of Alumina ore of Aluminium:
Hoope’s refining process used for electrolytic refining of Aluminium:
Wednesday, April 13, 2022
Chemical methods of purification/refining of metals:
Pudding process : Used for the manufacture of wrought iron from cast iron:
Vapour phase refining : Van Arkel–De Boer process :
Vapour phase refining : Mond's process:
Electrolytic refining process : used for Cu , Ni and Al :
Parting process : removal of Zn and Pb from Gold which is obtained by MacArthur-Forrest cyanide process:
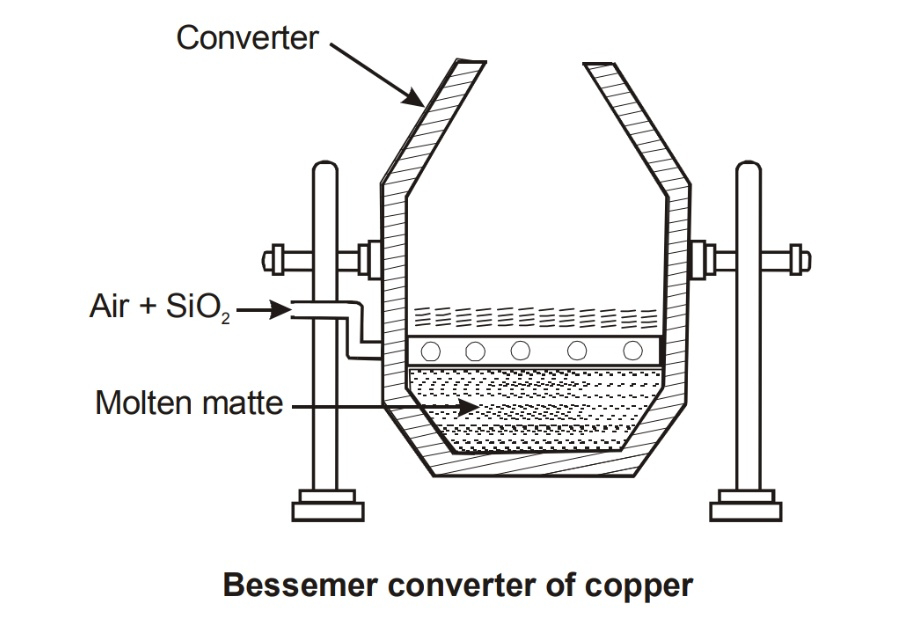
Bessemerisation : Purification of iron from cast Iron or pig iron :
Cupellation : lead (Pb) present in Silver removed by Cupellation process:
Polling process: This process is used for the purification of copper and tin.
Kroll's Process and Imperial metal industries (IMI) Process of reduction;
Parke's Process : Desilverisation of lead ;
The removal of the impurities of Ag from the commercial lead is called desilverisation of lead and is done by Parke's process . Thus, Parke's process is the desilverisation of lead.
In Parke's process, the commercial lead, which contains Ag as impurities, is melted in iron pots and 1% of Zn is added to it. The molten mass is thoroughly agitated. Since Ag is about 300 times more soluble in Zn than in Pb, most of the Ag present in the commercial lead as impurity mixes with Zn, to form Zn–Ag alloy. When the whole is cooled, two layers are obtained. The upper layer contains Zn–Ag alloy in the solid state, while the lower layer has lead in the molten state.
This lead containsonly 0.0004% of Ag and hence is almost pure. Lead obtained after removing most of Ag from it (desilverisation of lead) by Parke's process, is called desilverised lead. This lead contains the impurities of metals like Zn, Au, Sb etc. These metal impurities are removed from desilverised lead by Bett's
electrolytic process.
Zn–Ag alloy, formed in the upper layer, is skimmed off from the surface of the molten lead by perforate ladles. This alloy contains lead as impurity. This impurity of Pb is removed from the alloy by liquation process, in which Zn–Ag alloy is heated in a slopping furnace, when the impurity of Pb melts and hence drains away from the solid alloy. Thus purified Zn–Ag is obtained. Now Ag can be obtained from this purified Zn–Ag alloy by distillation process, in which the alloy is heated strongly in presence of little carbon in a fire–clay retort. Zn, being more volatile, distills off while Ag remains in the retort, carbon used in the process reuses the oxide of Zn, if formed. Ag obtained from Zn–Ag alloy is contaminated with a little of Pb as impurity. This impurity of Pb placed in a cupel (cupel is a boat–shaped) dish made of bone ash which is porous in nature) in a reverberatory furnace and heated in the presence of air. By doing so, lead (impurity) is oxidised to PbO(litharge) which volatilises and pure Ag is left behind in the cupel. Last traces of PbO are absorbed by the porous mass of the cupel.