We know that the Ligands which cause large
degree of crystal filed splitting are termed as strong field ligands. and Ligands that
cause only a small degree of crystal filed splitting are termed as weak field ligands.
The common ligands can be arranged in ascending order of crystal field
splitting energy. The order remains practically constant for different metals
and this series is called the spectrochemical series.
The spectrochemical series is an experimentally determined
series.
It is difficult to explain the order
as it incorporates both the effect of sigma
and Pi-bonding. The halides
are in the order expected from electrostatic effects. In other cases, we must
consider covalent bonding to explain the order. A pattern of increasing sigma- donation is as follows:
C-donors > P-donors
> N-donors > O-donors >S-donors > X-donors
The crystal field stabilization
produced by the strong CO and CN- is almost double that of halide
ions. This is attributing pi- bonding in which the metal donates electrons from
a filled t2g orbital
into a vacant orbital on the ligand. In a similar way, many unsaturated N
donors and C donors may also act as pi- acceptors.
Important Note:
Strong field ligands affects electronic
configuration of central metal my making
unpaired electron to pair up, while weak field ligands does not affect
electronic configuration of central metal atom i.e. they does not make unpaired
electrons to pair up.
Above Statement is valid for first
transition series however for second and third transition series unpaired
electrons, pair up irrespective of nature of ligands provide pairing
of electrons is allowed.

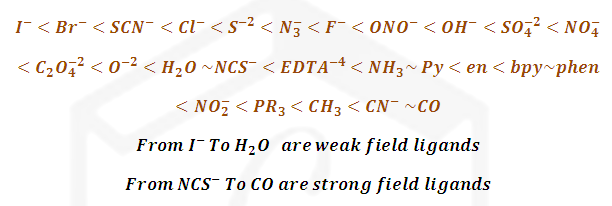

No comments:
Post a Comment