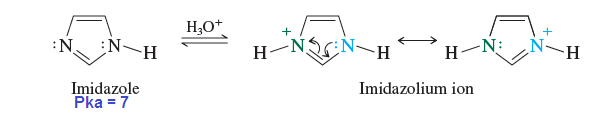We know that aliphatic amines are more basic than aromatic amines due to non delocalized of lone pair of nitrogen atom hence piperidine (option IV) is more basic than others
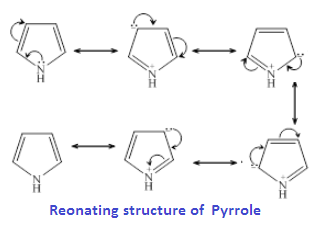
Pyridine is more basic than aniline and pyrrol because is lone pair of nitrogen is does not involved in pi cloud formation that means it is localised while pryrrol is less basic than aniline because is lone pair involved in pi cloud formation of pyrrol ring
Hence correct basic order is (iV) >(iii)>(I)>(ii)
Related Questions: