Search This Blog
Monday, April 20, 2020
The ammonia prepared by treating ammonium sulphate with calcium hydroxide is completely used by NiCl2.6H2O to form a stable coordination compound. Assume that both the reactions are 100% complete. If 1584 g of ammonium sulphate and 952g of NiCl2.6H2O are used in the preparation, the combined weight (in grams) of gypsum and the nickel-ammonia coordination compound thus produced is___. (Atomic weights in g mol–1: H = 1, N = 14, O = 16, S = 32, Cl = 35.5, Ca = 40, Ni = 59) (JEE-ADVANCED 2018 PAPER-2)
Galena (an ore) is partially oxidized by passing air through it at high temperature. After some time, the passage of air is stopped, but the heating is continued in a closed furnance such that the contents undergo self-reduction. The weight (in kg) of Pb produced per kg of O2 consumed is______ . (Atomic weights in g mol–1 : O = 16, S = 32, Pb = 207). (JEE-Advanced 2018 Paper-2)
Friday, January 31, 2020
A sample of impure Iron pyrite (FeS2) when 13.9 gm heated then it produces iron (iii) oxide (Fe2O3) and Sulphur dioxide (SO2). If 8.02 gm Iron (iii) oxide is obtained, what was the % purity of given sample (original-FeS2)?
When dilute HCl is added to 5.73 gm of contaminated CaCO3, 2.49 gm of CO2 is obtained. Find % purity of CaCO3 sample?
The Mass of 80% pure H2SO4 required to completely neutralize 106 gm of Na2CO3?
When 1.25 gm sample of Chalk is strongly heated. 0.44 gm of CO2 is produced. Calculate % purity of Chalk sample?
Calculate the amount of 80 % pure NaOH sample required to react with 21.3 gm Chlorine in hot condition.
Related Questions:
200 gm of CaCO3 sample on heating produces 11.2 liters of CO2 (g) at STP. Calculate % purity of CaCO3 sample?
Tuesday, April 2, 2019
Principle of atom conservation (PAOC):
We know that, in a chemical reaction, the atoms of each element remain conserved. If atoms are conserved, moles of atoms will also be conserved. This is known as the principle of atom conservation.
Total number of moles of an atoms of an element on reactant side = Total number of moles of atoms of an on product side.
Illustration
of POAC: Take a chemical
reaction as example.
(A)
According to the principle of atom conservation (POAC) for K atoms:
Total moles of K atoms in
reactant = total mole of K atoms in products
or
Moles
of K atoms in KClO3 = moles of K atoms in KCl.
Now, since 1 molecule of KClO3 contains 1 atom of K, 1mole of KClO3 contains 1 mole of K similarly, 1mole of KCl contains 1mole of K.
Thus, mole of K atoms in
KClO3 = 1 × moles of KClO3
And mole of
K atoms in KCl = 1 × moles of KCl
Hence
1 × moles of KClO3 =
1 × moles of KCl
(B) Similarly applying the principle of atom conservation for O atoms:
Mole of O in KClO3 = 3 ×moles of KClO3
And
moles of O in O2 = 2 × moles of O2
3
× moles of KClO3 = 2 × moles of O2
Illustrative
example (1): Write the POAC equations for all the atoms in the following reaction.
Illustrative solution:
Applying
POAC on (P) atoms:
P à H3PO4
Numbers of
moles of (P) atoms in P4 = Numbers of moles of (P) atoms in H3PO4
4x nP4= 1x nH3PO4
Applying
POAC on (H) atoms:
1x nHNO3
= 3 x nH3PO4 + 2 x nH2O
Applying
POAC on (N) atoms:
1 x nHNO3
= 1 x nNO2
Applying
POAC on (O) atoms:
3 x nHNO3
= 4 x nH3PO4 +2 x nNO2 + 1nH2O
Related Questions:
Illustrative example (2) 27.6 gm K2CO3 (138) was treated by a series of reagents so as to convert all its carbon into K2Zn3[Fe(CN)6]2(MM=698) Calculate mass of K2Zn3[Fe(CN)6]2.
Illustrative example (3)0.32 gm mole of LiAlH4 in ether solution was placed in a flask and 74 gm (1 mole ) of t- butyl alcohol was added . The product is LiAlHC12H27O3. Calculate mass of product if all lithium atoms are converted.
Illustrative example (4) All the carbon atom present in KH3(C2O4)2.2H2O weighting 254 gm is converted to CO2. How many gram of CO2 were obtained.
Monday, April 1, 2019
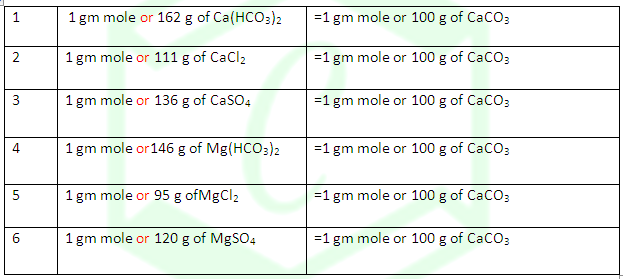
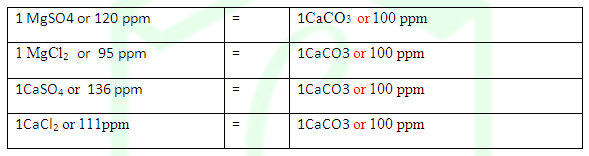
DEGREE OF HARDNESS OF HARD WATER:
Friday, March 15, 2019
EUDIOMETRY-VOLUME-VOLUME ANALYSIS OF GAS:
The edudiometric relationship amongst gases, when they react with one another, is governed by two laws, they are illustrated as, Gay-Lussac law and Avogadro’s law.
(1) Gay-Lussac Law:
According to Gay Lussac’s law, the volumes of gaseous reactants reacted and the volumes of gaseous products formed, are always bear a simple ratio at same temperature and pressure.
According Avogadro law A samples of different gases which contain the same number of molecules occupy the same volume at the same temperature and pressure. This law is also known as Avogadro’s hypothesis and given by Amadeo Avogadro in 1812.
Gaseous reactions are studied in a closed graduated tube open at one end and the other closed end of which is provided with platinum terminals for the passage of electricity through the mixture of gases, Such a tube is known as Eudiometric tube and hence the name Eudiometry also used for “Volume analysis of Gas”.
During Gas analysis, the Eudiometer tube filled with mercury is inverted over a trough containing mercury. A known volume of the gas or gaseous mixture to be studied is next introduced, which displaces an equivalent amount of mercury. Next, a known excess of oxygen is introduced and the electric spark is passed, whereby the combustible material gets oxidized.
O3 -- Turpentine oil
O2 - Alkaline pyrogallol
NO - FeSO4 solution
Cl2, SO2, CO2 -- Alkali solution NaOH, KOH, etc.
NH3, HCl - Water
H2O, - CuSO4, CaCl2
CO – Ammonical Cuprus chloride solution (NH4OH+ Cu2Cl2 )
(1) Molecular formulae of Gaseous Hydrocarbons.
(2) The composition of Gaseous mixtures.
(3) Molecular formulae of Gases.
(4) Volume-volume relationship Gaseous reactions.
SOLUTION:
Sunday, March 10, 2019
GRAVIMETRY- WEIGHT-WEIGHT ANALYSIS:
(I) Silver ppt with halides ions
(II) Calcium ppt with Oxalates
(III) Barium ppt with Sulphates
ILLUSTRATIVE EXAMPLE (2): A mixture of FeO and Fe2O3 is produced by 1 mole of Fe reacts Completely with 0.65 moles of O2. Find out the mole’s ratio of ferrous oxide to ferric oxide.?
ILLUSTRATIVE EXAMPLE (3): 8 gm mixture of sodium carbonate and sodium bicarbonate are heated until a constant mass equal to 6 gm is obtained calculate % of sodium bicarbonate in the original mixture ?.
ILLUSTRATIVE EXAMPLE (4): 1 litre of a mixture of CO and CO2 is passed through a tube containing red hot charcoal. The total volume of the mixture become 1.5 litre. Calculate Volume % of CO in the original mixture.?
ILLUSTRATIVE EXAMPLE (5): 1-gram mixture of cuprous and cupric oxide was reduced to 0.839 gm metallic cupper. What is the weight of cuprous oxide in given sample.? (Cu= 63.5, O= 16)
ILLUSTRATIVE EXAMPLE (6): A mixture of pure AgCl and AgBr contains 60.94 % silver by mass, what is percentage of AgCl in sample.? (Ag= 108, Br= 80 Cl = 35.5)
ILLUSTRATIVE EXAMPLE (7): 1.48 gm mixture of CaCO3 and MgCO3 was heated to a constant weight till 0.96 gm residue formed, % of MgCO3 in the sample was?
ILLUSTRATIVE EXAMPLE (8): When 40 gm of 200% solution by weight was cooled, 50 gm of solute precipitated. The percentage of concentration of remaining solution is.
ILLUSTRATIVE EXAMPLE (9): 18.4 gm mixture of CaCO3 and MgCO3 was heated to produce a constant volume of carbon dioxide is 4.48 litre the calculate amount of MgCO3 and CaCO3 in mixture?
ILLUSTRATIVE EXAMPLE (10): 31.3 gm NaCl mixture of NaBr and NaCl treated with H2SO4, 4.48 gm of Na2SO4 is produced. Then Calculate the amount of NaCl and NaBr in mixture.
Friday, March 1, 2019
CONCEPT OF LIMITING REAGENTS:
HOW TO FIND LIMITING REAGENT:
(1) Divide the given moles of each reagent by its Stoichiometric coefficient.
(2) The reagent for which the division comes out minimum is called Limiting reagent.
SIGNIFICANT OF LIMITING REAGENT:
(1) The amount of product is decided by Limiting reagent .
(2) Amount of excess reagent left over and participate in the reaction is decided by Limiting reagent.
GENERAL ILLUSTRATION:
Carried out by taking 24 gm of Carbon (2 moles ) and 96 gm (3 moles ) of oxygen (O2) find
(2) How many moles of CO formed
(3) percentage (%) of reagent left over .
(1) Find the limiting reagent
(2) Mass of Li2O produced in reaction
(3) % of excess reagent consumed
(4) % of excess reagent undergoes in reaction.