Search This Blog
Sunday, May 24, 2020
B-F bond length in BF3 is shorter than B-F bond length in (BF4)- why?
Thursday, February 13, 2020
What is structure of solid Ortho Boric acid ?
Related Questions:
What is the structure of trimetaboric acid and trimetaborate ion?
Related Questions:
Monday, February 10, 2020
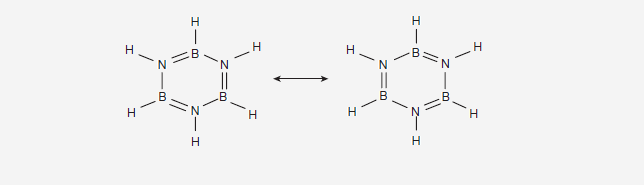
Why Borazine is more reactive than benzene towards Electrophic Aromatic substitution reactions ?
Sunday, February 9, 2020
Why Borazine (B3N3H6) is also known as inorganic benzene ?.
Related Questions:
Why Ga has small size than Al exceptionally
Why aqueous solution of borax reacts with two moles of acids ?
What is structure of solid Ortho Boric acid ?
What is the structure of trimetaboric acid and trimetaborate ion?
Why Borazine is more reactive than benzene towards Electrophic Aromatic substitution reactions ?
Why Borazine (B3N3H6) is also known as inorganic benzene ?.
Why B-F bond length in BF3 is shorter (130 pm) than B-F bond Iength in BF4- (143 pm)?. Explain.
Why B-F do not exist as dimer?. Explain.
Although anhydrous aluminium chloride is covalent but its aqueous solution is ionic in nature. Why?
Why Boric acid become strong acid in the presence of cis 1,2-diol or 1,3-diol ?Why B-F bond length in BF3 is shorter (130 pm) than B-F bond Iength in BF4- (143 pm)?. Explain.
BF3 has a planar trigonal geometry due to sp2 hybridisation of boron in BF3. Boron atom in BF3 is electron defficient (incomplete octet) and fluorine atom is electron rich (due to the presence of three lone pair of electrons on fluorine atom).
Boron and fluorine both belong to the same
period. Hence their bonding orbitals have almost same size and same
energies.Thus Back Bonding takes place between B and
F. This introduces a partial double
bond character between B-F of BF3. Thus B-F bond length decreases from
normal B-F bond length.
In
BF4-,the hybridization
of boron is sp3. The B-F bond has pure single bond character. Thus B-F bond
length in BF4-, is more than the B-F bond length in BF3
.
Why aqueous
solution of AlCl3 is acidic in nature ?
What happen when
aq AlCl3 react with Acid or Base?
Although
anhydrous aluminium chloride is covalent but its aqueous solution is ionic in
nature. Why?
Why BF3 do not
exist as dimer?. Explain.
Why B-F bond
length in BF3 is shorter (130 pm) than B-F bond Iength in BF4- (143 pm)?.
Explain.
B-F bond length
in BF3 is shorter than B-F bond length in (BF4)- why?
What is product
of reaction between diborane (B2H6) and ammmonia (NH3)?
Why methylation
of Diborane (B2H6) replace four hydrogen only ?
What is use of
Orthoboric acids?
What is basicity
of "Boric acid" ?
Why Boric acid
exist in solid state ?
What is
structure of solid Ortho Boric acid ?
What is effect
of heat on Borax?
What is the
structure of trimetaboric acid and trimetaborate ion?
What is the
Sodium per borate ,give the structure and its uses?
Why aqueous
solution of borax reacts with two moles of acids ?
What is the
molecular formula of Borax ?
Why Boric acid
become strong acid in the presence of cis 1,2-diol or 1,3-diol ?
Why Borazine is
more reactive than benzene towards Electrophic Aromatic substitution reactions
?
Why Borazine
(B3N3H6) is also known as inorganic benzene ?.
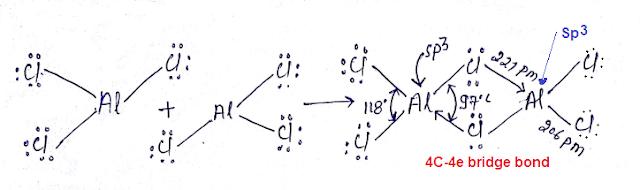
Four-center
two-electron bond (4C-2e Bond): Structure of AlCl3:
Why BF3 do not exist as dimer?. Explain.
Related Questions:
Why Ga has small size than Al exceptionally
Why aqueous solution of borax reacts with two moles of acids ?
What is structure of solid Ortho Boric acid ?
What is the structure of trimetaboric acid and trimetaborate ion?
Why Borazine is more reactive than benzene towards Electrophic Aromatic substitution reactions ?
Why Borazine (B3N3H6) is also known as inorganic benzene ?.
Why B-F bond length in BF3 is shorter (130 pm) than B-F bond Iength in BF4- (143 pm)?. Explain.
Why B-F do not exist as dimer?. Explain.
Although anhydrous aluminium chloride is covalent but its aqueous solution is ionic in nature. Why?
Why Boric acid become strong acid in the presence of cis 1,2-diol or 1,3-diol ?Saturday, February 8, 2020
Although anhydrous aluminium chloride is covalent but its aqueous solution is ionic in nature. Why?
Aluminium forms covalent
compound with chloride because lonisation enthalpy (∆iH= +5137 kJ/mole) of Aluminium is very high due to small size
and chlorine is unable to convert Al into Al+3 ions.
However, when
anhydrous AlCl3 (which is covalent in character) is
dissolved in water, it undergoes hydration as follow:
Al2CI6 + H2O --> 2[Al(H2O)6]+3 + (∆H)
Hydration of anhydrous aIuminium chloride is highly exothermic in nature. The hydration enthalpy is more than ionisation enthalpy of aluminium.This hydration enthalpy removes all the three valence electrons of the aluminium leading to the formation of Al3+ more easly.This AI3+ is hydrated with water and form a complex ion. Thus in water Al exist as [Al(H2O)6]+3 . The three electrons of aluminum is accepted by CI of AlCl3. Thus hydrated AlCl3 represented [Al(H2O)6]Cl3 and it is ionic in nature.
Related Questions:
(1) Although anhydrous aluminium chloride is covalent but its aqueous solution is ionic in nature. Why?
(2) Why Ga has small size than Al exceptionally
(3) Why aqueous solution of borax reacts with two moles of acids ?
(4) What is structure of solid Ortho Boric acid ?
(5) What is the structure of trimetaboric acid and trimetaborate ion?
(6) Why Borazine is more reactive than benzene towards Electrophic Aromatic substitution reactions ?
(7) Why Borazine (B3N3H6) is also known as inorganic benzene ?.
(8) Why B-F bond length in BF3 is shorter (130 pm) than B-F bond Iength in BF4- (143 pm)?. Explain.
(9) Why B-F do not exist as dimer?. Explain.
(10) Although anhydrous aluminium chloride is covalent but its aqueous solution is ionic in nature. Why?
(11) Why Boric acid become strong acid in the presence of cis 1,2-diol or 1,3-diol ?
(12) Four-center two-electron bond (4C-2e Bond): Structure of AlCl3:
(13) What is the molecular formula of Borax ?
(14) What is the difference between the structure of AlCl3 and diborane?
Saturday, January 25, 2020
Why Boric acid become strong acid in the presence of cis 1,2-diol or 1,3-diol ?
Monday, December 30, 2019
What is the molecular formula of Borax ?
Three-center four-electron Bridge bond (3C-4e Bond): Structure of AlCl3:
Related Questions:
(1) Why aqueous solution of AlCl3 is acidic in nature ?
(2) What happen when aq AlCl3 react with Acid or Base?
(3) Although anhydrous aluminium chloride is covalent but its aqueous solution is ionic in nature. Why?
(4) Why BF3 do not exist as dimer?. Explain.
(5) Why B-F bond length in BF3 is shorter (130 pm) than B-F bond Iength in BF4- (143 pm)?. Explain.
(6) B-F bond length in BF3 is shorter than B-F bond length in (BF4)- why?
(8) What is product of reaction between diborane (B2H6) and ammmonia (NH3)?
(9) Why methylation of Diborane (B2H6) replace four hydrogen only ?
(10) What is Use of Boric Acid?
(11) What is use of Orthoboric acids?
(12) What is basicity of "Boric acid" ?
(13) Why Boric acid exist in solid state ?
(14) What is structure of solid Ortho Boric acid ?
(15) What is effect of heat on Borax?
(16) What is the structure of trimetaboric acid and trimetaborate ion?
(17) What is the Sodium per borate ,give the structure and its uses?
(18) Why aqueous solution of borax reacts with two moles of acids ?
(19) What is the molecular formula of Borax ?
(20) Why Boric acid become strong acid in the presence of cis 1,2-diol or 1,3-diol ?
(21) Why Borazine is more reactive than benzene towards Electrophic Aromatic substitution reactions ?
(22) Why Borazine (B3N3H6) is also known as inorganic benzene ?.
(23) Four-center two-electron bond (4C-2e Bond): Structure of AlCl3:
(24) What is the difference between the structure of AlCl3 and diborane?
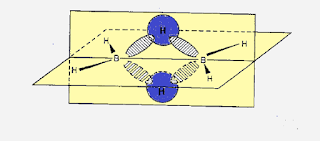
What is the difference between the structure of AlCl3 and diborane?
Related Questions:
(1) Why aqueous solution of AlCl3 is acidic in nature ?
(2) What happen when aq AlCl3 react with Acid or Base?
(3) Although anhydrous aluminium chloride is covalent but its aqueous solution is ionic in nature. Why?
(4) Why BF3 do not exist as dimer?. Explain.
(5) Why B-F bond length in BF3 is shorter (130 pm) than B-F bond Iength in BF4- (143 pm)?. Explain.
(6) B-F bond length in BF3 is shorter than B-F bond length in (BF4)- why?
(8) What is product of reaction between diborane (B2H6) and ammmonia (NH3)?
(9) Why methylation of Diborane (B2H6) replace four hydrogen only ?
(10) What is Use of Boric Acid?
(11) What is use of Orthoboric acids?
(12) What is basicity of "Boric acid" ?
(13) Why Boric acid exist in solid state ?
(14) What is structure of solid Ortho Boric acid ?
(15) What is effect of heat on Borax?
(16) What is the structure of trimetaboric acid and trimetaborate ion?
(17) What is the Sodium per borate ,give the structure and its uses?
(18) Why aqueous solution of borax reacts with two moles of acids ?
(19) What is the molecular formula of Borax ?
(20) Why Boric acid become strong acid in the presence of cis 1,2-diol or 1,3-diol ?
(21) Why Borazine is more reactive than benzene towards Electrophic Aromatic substitution reactions ?
(22) Why Borazine (B3N3H6) is also known as inorganic benzene ?.
(23) Four-center two-electron bond (4C-2e Bond): Structure of AlCl3:
(24) What is the difference between the structure of AlCl3 and diborane?