Orthoboric acid, H3BO3,
commonly known as boric acid, and Metaboric acid, HBO3 , are two
common oxy acids of boron . Orthoboric acid
naturally found in volcanic steam vents called Suffioni.
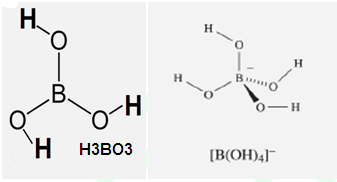
STRUCTURE OF BORIC ACID:
(1) In
dilute Solution Boric acid exist as monomeric form;
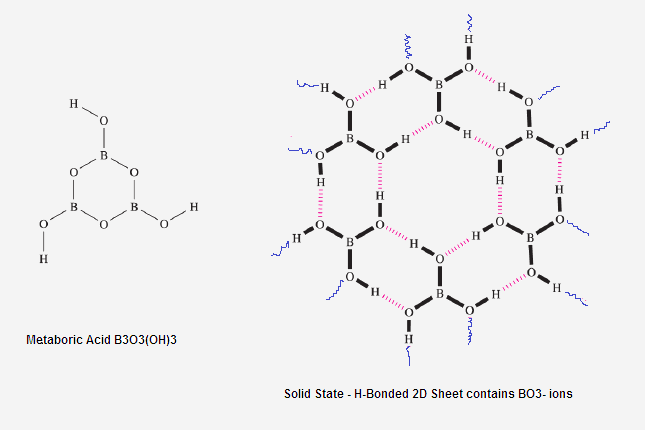
(2) When concentration of the solution
of acids is very high then boric acid
exist as polymeric metaboric and many more ions.
(3) In Solid State It exist sp3
hybridised BO3- 2D Sheet .
GENERAL PROPERTIES:
(1) H3BO3 is soluble in water and behaves as weak monobasic
acid. It does not donate protons but rather it accepts OH-
.Therefore it acts as a Lewis acid [B(OH)3]
.It is not a proton donor because it accept lone pair or hydroxyl ion from
water.
(2)
Since B(OH)3 only partially reacts with water to form H3O+ and [B(OH)4]- it behaves as a weak acid. Thus it
cannot be titrated satisfactorily with NaOH as a sharp end point is not
obtained.
(3)
B(OH)3 (Boric acid ) does not
titrated even strong alkali like NaOH
but If certain polyhydroxy compounds such as
glycerol, mannitol or sugar are added to the titration mixture then B(OH)3
behaves as a strong monobasic acid and hence can be titrated with NaOH and end
point is diluted using phenolphthalein as
indicator.
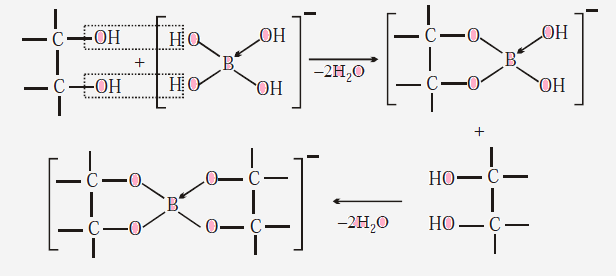
(4)
The added compound must be a cis-diol to enhance the acidic proprieties. In
this way the cis-diol forms very stable complexes with [B(OH)4]¯ formed in
forward direction above, thus effectively removing it from solution. Hence
reaction proceeds in forward direction (Le-Chatelier principle.)
PHYSICAL PROPERTIES:
CHEMICAL PROPERTIES:
USES:
Related Questions:
Reference Books:
The most recommended reference books of Inorganic chemistry for IIT JEE (Mains/Advanced) and NEET exams are given here. you can by this book from Amazon from following links:


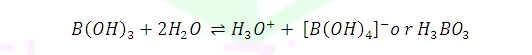
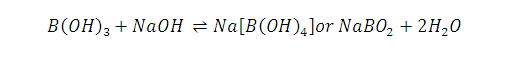
No comments:
Post a Comment