Metallic radius or Crystal radius:
(1) The term crystal radius is used to denote the
size of atoms in metal.
(2) Metal atoms are closely packed spheres in
metallic crystal. The metal atoms are supposed to touch one another in crystal.
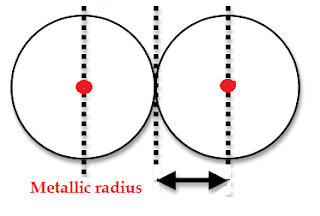
(3) Metallic radius is defined as one half the distances
between the centres of the nuclei of two atoms in a metallic crystal.
(4) Metallic radius is determined by X-ray diffraction
method.
(5) Metallic radii are about 10 to 15 % higher
than the single bond covalent raddi of those elements. Thus single bond covalent radius is smaller than the metallic
radius due to the no overlapping of atomic orbital in metallic bond.
Vander Waal’s radius > Metallic radius > Covalent radius
(6) For the
simplicity the term atomic radius is used for covalent radius as well as
metallic radius depending on whether the element is a non-metal or metal. However,
the atomic radii of inert gases are expressed in the terms of Vander Waal’s
radii.
(7) Metallic
radius is inversely proportional to the metallic bond strength.
(8) More
metallic radius –loose crystal packing-less bond strength. (BCC)
(9) Less
metallic radius –Tight crystal packing (FCC) - high bond strength.(HCP)
(10) For
non-metal, atomic radius means covalent radius.
(11) For metal,
atomic radius means metallic radius.
(12) For inert gases, atomic radius means Vander Waal’s radius.


No comments:
Post a Comment