For maximum possible labbelling amount of SO3 is 100 gm, It means 100 gm Oleum sample contains 100 gm SO3 only and zero( 0 )gm H2SO4 ,hence maximum labbelling possible is 122.5 %.
Search This Blog
Sunday, October 11, 2020
Saturday, October 10, 2020
Why Oleum is known as fuming Sulphuric acid?
(1) Oleum can be represented by the formulaySO3.H2O where y is the total molar sulphur trioxide content .the value of y can be varied to different Oleum sample.
(2) Oleum also be expressed as H2SO4.xSO3where x is molar free suphur trioxide.
(3) Oleum is the solution of of sulphur trioxide in sulphuric acid , it is also known as fuming sulphuric acid or Pyrosulphuric acid(H2S2O7=H2SO4+SO3).
(4) Oleum sample contain two type of SO3.
(2) Oleum also be expressed as H2SO4.xSO3where x is molar free suphur trioxide.
(3) Oleum is the solution of of sulphur trioxide in sulphuric acid , it is also known as fuming sulphuric acid or Pyrosulphuric acid(H2S2O7=H2SO4+SO3).
(4) Oleum sample contain two type of SO3.
(i) Free SO3:- It is that SO3 which combined with water water to give H2SO4 .
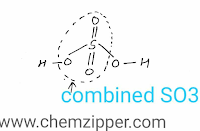
(ii) Combined SO3:- The SO3 which is present in H2SO4 itself is known as combined SO3 and it does not react with water.
Subscribe to:
Comments (Atom)