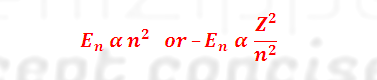Relation between Total energy (TE), Kinetic energy (KE) and Potential energy (PE):
Important conclusions:
(1) The minus sign for the energy of an electron in an orbit represents attraction between the +vely charged nucleus and negatively charged electron.
(2) Energy of an electron at infinite distance from the nucleus is zero.
(3) As an electron approaches the nucleus, the electrical attraction increases, energy of electron decreases and it becomes negative.
(4) Energy of an electron increases as the value of ‘n’increases i.e.
(5) Value of ‘n’ remaining unchanged, the amount of energy associated with an electron remains unaltered.
(6) Energy of electron in first, second, third and fourthorbit are –13.6, –3.4, –1.5, and –0.85 eV/atomrespectively.
(7) Although the energy of electron increases with increase in the value of ‘n’ (orbit), yet the difference of energy between successive orbits decreases. Thus E2 – E1 > E3 – E2 > E4 – E3 > E5 – E4 >, etc…