Search This Blog
Thursday, November 25, 2021
Tuesday, November 23, 2021
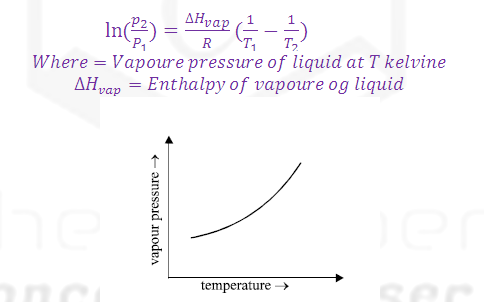
Class Claperon equation:
Vapour pressure is directly proportional to the Temperature so that on increasing temperature the rate of evaporation increases and rate of condensation decreases and hence vapour pressure increases.
The dependence of vapour pressure and temperature is given by CLASIUS CLAPERON equation.
Related topics:
Thursday, November 18, 2021
How many of the elements exist as gases at 25°c?
H,N,O, flourine (F), Chlorine (Cl),He, Neone (Ne) , Argone (Ar) kriptane (Kr) , Xenone (Xe) these are the elements found in gaseous state at 25℃ present in periodic table.(2) What are the Amphoteric metals ? gives Examples.
(4) What is Mendeleev's periodic table ? give important features and draw back of Mendeleev's table.
(5) What is atomic density ? give the periodicity of atomic density in periods and groups.
(6) What is atomic volume ? and what is periodicity of atomic volume in groups and periods ?
(7) Why there are 2, 8 and 8 elements in first, second and third periodic of periods table respectively ? Explain.
(8) In alkali metal group which is the strongest reducing agent in aqueous solution and why?
(9) The electron affinity of sulphur is greater than oxygen. Why?
Related Questions:
(1) Total numbers of elements which are liquid at normal temperature is ?
(2) What are the Amphoteric metals ? gives Examples.
(3) Name of total metalloids present in periodic table ?
(4) What is Mendeleev's periodic table ? give important features and draw back of Mendeleev's table.
(5) What is atomic density ? give the periodicity of atomic density in periods and groups.
(6) What is atomic volume ? and what is periodicity of atomic volume in groups and periods ?
(7) Why there are 2, 8 and 8 elements in first, second and third periodic of periods table respectively ? Explain.
(8) In alkali metal group which is the strongest reducing agent in aqueous solution and why?
(9) The electron affinity of sulphur is greater than oxygen. Why?
(10) The first ionization energy of carbon atom is greater than that of boron atom, whereas reverse is true for the second ionization energy. Explain.
Monday, November 15, 2021
Liquid A and B forms an ideal solution and initial vapour pressure of A and B are P°A= 0.4 atm and P°A= 0.6 atm respectively. In a cylinder piston arrangement, 2 mole vapour of A and 3 mole vapour of B is collected at 0.42 atm.
(1) predict wheather the vapour will condense or not ?
(2) If the vapours are compared slowly and isothermally at what pressure F drop of liquid will form?
(3) If initial volume of vapour was 10 litre , at what volume first drop of liquid is form?
(4) What is the composition of first drop of liquid formed?
(5) If vapour pressure are further compressed. At what pressure almost complete condensation of vapour will occure?
(6) Determine the composition of last traces of vapours remains?
(7) What is the composition of system at 0.55 atm pressure?
(8) Determine the composition of liquid and vapour formed at 0.51 atm also calculate the moles of liquid of A and B in liquid and vapour formed ?
(9) At what pressure half of the total amount of vapour will condense?
Related Questions;
Subscribe to:
Posts (Atom)