Heat is defined as the energy
that flow into or out of the system because of a difference in temperature between
system and surrounding.
HEAT CAPACITY(Q)
Total degree of freedom = 3N (Where N is Number of atom in molecule)
Total degree of freedom:= ftrans + f rot + f vib and fvib= 3N- ftrans + f rot
Note; Work is more organised way
of energy transfer as compared to hear exchange.
IUPAC Sign convention of Heat: sign of heat will negative (-Ve) if
heat is released by the system given by system while sign of heat will be
positive (+Ve) if heat is given to the system.
(i) qV = nCvdT (for constant volume
process)
(ii) qp = nCpdT (for constant
pressure process)
(iii) Cp,m – Cv,m = R
(iv) Cv & Cp depends on
temperature even for an ideal gas.( C = a + bT + cT2 .....)
(v) It is a path function
Cv, Cp are heat capacity of system
and Cv,m, Cp,m are heat capacity of one mole system at
constant volume and pressure
respectively.
“Exchange of heat and work(P–V) in
between system and surrounding always
occur through boundry of system”
Note: heat exchange can be measured with the help of Heat
Capacity.
We know that usually on increasing in
temperature is proportional to the heat transfer
q=coefficient x ∆T
The coefficient depend on the size,
composition and nature of the system so we can also write it as
q=C∆T
where C is called Heat capacity
The heat capacity (C):
q=C∆T or
q=C∆T or
C= q/∆T unit- J/K
If ΔT=10=1K
Then C=q
It is equal to amount of heat needed
to raise the temperature of the sample of any substance by one degree Celsius
(or Kelvin).
heat
Capacity depend on quantity, nature as
well as physical state of the system. And the heat capacity is extensive. it may be made intensive as specific heat capacity
Specific Heat Capacity(Cs ):
q=Csm∆T or
q=Csm∆T or
Cs= q/m∆T
If ΔT=10=1K and m = 1g
Then
Cs=q
It is equal to amount of heat required to raise the temperature of 1gm
substance by one degree centigrade .it is intensive properties.
Molar Heat Capacity(Cm ):
q=Cmn∆T or
q=Cmn∆T or
Cm= q/n∆T
If ΔT=10=1K and n=1mole
Then Cm=q
It is equal
to amount of heat required to raise the temperature of 1 Mole substance by one
degree centigrade
EXAMPLE(1): The latent heat of fusion of ice at
0ºCis 80 cal/gm the amount of heat needed to convert 200 gm ice into water at
0ºC is ?
(A) 80 cal
(B) 16000 J (C) 16000 cal (D) 1600 cal
SOLUTION:
Ans (C) q = m.L = 200 × 80 = 16000 cal
EXAMPLE(2): Calculate the amount of heat required
to raise the temperature of 50 gm water from 25ºC to 55ºC.Specific heat
capacity of water = 4.2 J/ºC-gm.
(A) 126 J (B)
210 J (C) 6300 J (D) 1500 J
SOLUTION: Ans. (C)
q = m.s.ΔT = 50 × 4.2 × (55 – 25) = 6300 J
EXAMPLE (3): Five moles of a monatomic ideal gas is
heated from 300K to 400K at constant pressure. the amount of heat absorbed is :
(A) 500 cal (B) 1500 cal (C) 2500 cal (D) 2500 J
SOLUTION: Ans. (C)
qp = CpΔT = n.Cp,m ΔT
= 5 × 5/2× (400
– 300) = 2500 cal
EXAMPLE (4): 2
moles of an ideal gas absorbs 720 cal heat when heated from 27ºC to 87ºC, at
constant volume. 'ɤ' for the gas is :
(A) 1.5 (B) 1.4 (C) 1.6 (D) 1.33
SOLUTION :Ans. (D)
qv
= n.Cv,m. ΔT
Cv,m =qv/nΔT
=
720/2x(87 – 27)
= 6 cal/K-mol
Now, r = 1 +R/Cv,m
= 1 + 2/6
= 1.33
EXAMPLE (5): 500 gm ice at 0ºC is added in 2000 gm water
at tºC. If the final temperature of system is 0ºC, then the value of 't' is
(latent heat of fusion of ice = 80 cal/gm and specific heat capacity of water=cal/gm-ºC)
(A) 20 (B) 40
(C) 10 (D) 2
SOLUTION: Ans. (A)
Heat lost by water = heat gained by ice
or, (m.s. ΔT)water = (m.L)ice
or, 2000 × 1 × (t – 0) = 500 × 800
Δ t = 20ºC
EXAMPLE(6): What is the heat in Joules required to raise the temperature of 25 grams of water from 0 °C to 100 °C? What is the heat in calories?
(Given: specific heat of water = 4.18 J/g·°C)
SOLUTION: Use the formula q = mcΔT
where
q = heat energy
m = mass
c = specific heat
ΔT = change in temperature
q = 25gx4.18 J/g·°x(100 °C - 0 °C)
q = 25gx4.18 J/g·°Cx(100 °C)
q = 10450 J
where
q = heat energy
m = mass
c = specific heat
ΔT = change in temperature
q = 25gx4.18 J/g·°x(100 °C - 0 °C)
q = 25gx4.18 J/g·°Cx(100 °C)
q = 10450 J
We know 1 Calorie=4.18 J
So 10450 J in Calorie = 10450/4.8=2500 calorie
FOR LARGE HEAT CHANGE :
Q= nCm (T2-T1)
Case- (2) Cm = f(T)
Cm = a + bT+
cT2 +……..
Case- (3) The theoretical value of Cvm
and Cpm for Ideal gas can determined by using degree of freedom.
CHARACTERISTIC OF HEAT CAPACITY:
(1): The heat
capacity Of any system should depend upon temperature because by increasing temperature
of system different degree of freedom get excited.
(2): When
temperature approaches zero then heat absorbed by the solid mainly converted into vibration potential energy of molecule
resulting in very small increase in temperature, hence ‘C’ increases sharply
with increase in temperature.
Normally C Directly proportional T3
(3):
When the temperature at Melting point of solid ,then heat capacity becomes
nearly constant for solid elements.
Molar
heat capacity= 6.4 Cal/K mole
Or specific heat capacity x atomic
weight=6.4 (Dulong and petite’s law)
(4): Exactly
at melting point, the heat capacity become infinite as ΔT=0
(5):
the heat capacity of liquid is greater than that of solid because of rotational
degree of freedom also excited.
(6):
In liquid heat capacity also depend
upon temperature and also infinite at boiling point.
(7):
the heat capacity of gas become less than liquid because all vibrational and
rotational degree of freedom converted into translational degree of freedom.
(8):
the heat capacity of gases depend upon
their atomicity.
(9):
If the heat capacity depends upon temperature.
(10);
As heat (q) is path function, any substance may have infinite heat capacity.
Example
for any substance.
Isothermal process = infinite
Adiabatic
process = 0
Isobaric
process = Cp
Isochoric process = Cv
Normally ,we use Cp and Cv value as characteristic of substance.
DEDREE OF FREEDOM :
It is equal to number of modes of energy transfer when a gaseous molecule undergoes collision. OR
It represent the number of independent modes to describe the molecular motion.Total degree of freedom = 3N (Where N is Number of atom in molecule)
1-
Translational degree of freedom is 3 (three)
always for mono,di and tri atomic molecule.
2-
Rotational degree of freedom is zero
for mono atomic
gas,2 (two) for diatomic molecules and 3 (three)
for triatomic
molecule
3-Vibrational
degree of freedom is also zero for mono atomic gas and 1(one) diatomic gas molecule
and for polyatomic gases VDOF is
calculated individually.( fvib= 3N- ftrans+ frot)
Total degree of freedom:= ftrans + f rot + f vib and fvib= 3N- ftrans + f rot
Molecules N TDF
He 1 3
O2 2 6
CO2 3 9
NH3 4 12
PCl5 6
18
Case-1
Monoatomic Diatomic Triatomic
(linear) Triatomic (Non linear)
f total
=3 ftotal =6 ftotal =9 f total=9
f trans=3 ftrans =3 ftrans=3 ftrans=3
f
rot =0 frot =2
frot = 2 frot =3
f vib =0 fvib =1 fvib = 4 fvib =3
Q =n CmdT
QV=n CvmdT
Cvm=(dQ/dT)v
By FLOT dq=dU+dW and at constant volume
dW=0 so dQv=dU
Hence
LAW OF EQUIPARTIAL OF ENERGY :
Average energy associate with each molecule per degree of freedom is U= 1/2KT (where K is Boltz’s man constant.
Average energy associate with each molecule per degree of freedom is U= 1/2KT (where K is Boltz’s man constant.
Let degree of freedom is =
f then U is U=1/2fkT
And U=1/2fkTNA per molecule we know kNA=R
U=1/2fRT
and dU/dT=1/2fR
And dU/dT=Cv hence
Cv=1/2fR
Cv=1/2ftransR +1/2frotR (Where
Vib degree inactive in chemistry)
For ideal gas Cpm-Cvm=R
and Gama= Cpm/Cvm
Adiabatic exponent :Adiabatic exponent (Gama) for a mixture
of gas with different heat capacity is defined as :
where n1, n2 ........................ are moles of
different gases
Example:Calculate change in internal energy of
10 gm of H2 ,when it's state is changed from(300K, 1Atm) to (500 K,
2Atm) ?
Solution: For ideal gas
Cv for H2 (diatomic) in low temperature
range will be 5R as vibrational part is not included.




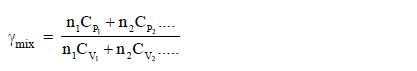

No comments:
Post a Comment