(1)The amount of hardness causing substances
(soluble salts of calcium or magnesium) in a certain volume of water measures
the extent of hardness or degree of hardness.
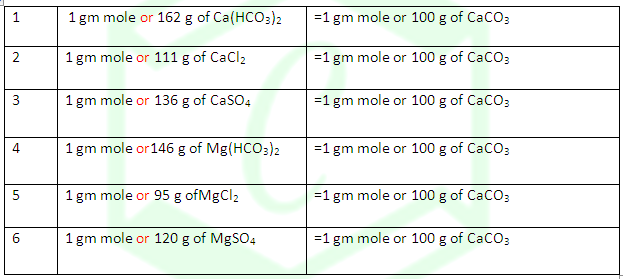
(2) Hardness of water is
always calculated in terms of calcium carbonate although this is never
responsible for causing hardness of water because of its insoluble character.
(3) The
reason for choosing CaCO3 as the standard for calculating hardness
of water is the ease in calculation as its molecular weight is exactly 100.Thus
the amount of various hardness causing substances in terms of CaCO3 can
be calculated on the basis of the following relations.
Thus the various types of harnesses in a water
sample may be calculated as below.
Temporary
hardness =Hardness due to Ca(HCO3)2 + Hardness due to Mg(HCO3)2
Permanent hardness = Hardness due to CaCl2 + due
to CaSO4 + due toMgCl2 + due toMgSO4
Degree of hardness is
usually expressed as parts per million (ppm)
and thus may be defined as the number of parts by weight of CaCO3 (equivalent
to calcium and magnesium salts) present in a million (106) parts by weight
of water.
From the
above definition, we can say that;
ILLUSTRATIVE EXAMPLE (1):
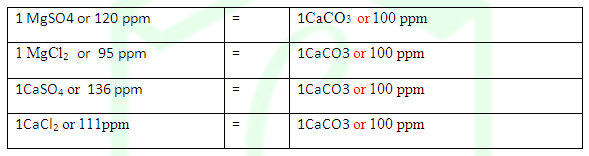
Determine the degree of hardness of a sample of water containing 30 ppm of MgSO4.
SOLUTION: 1 MgSO4
=1CaCO3
120 ppm = 100 ppm
Hence 30 ppm = 25 ppm Ans


No comments:
Post a Comment