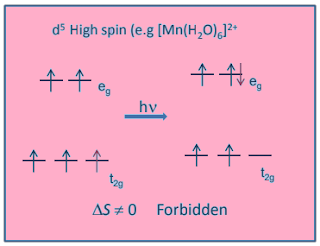
There are 5
unpaired electrons in Mn+2 ion and we know that.
Spin multiplicity is = (2S+1)
Where S is some of spin thus S = 5×1/2=5/2
Hence multiplicity = (2S+1)= 6
Spin multiplicity is = (2S+1)
Where S is some of spin thus S = 5×1/2=5/2
Hence multiplicity = (2S+1)= 6
we know multiplicity of excited state cannot be six, thus electronic transition in Mn+2 are spin forbidden, hence [Mn(H2O)6]+2 salt appear colourless.
Also [Mn(H2O)6]+2
has centre of symmetry and
in such cases electronic transition are expected to be laporate forbidden.

No comments:
Post a Comment