Aspirin
is the most well known member of a group of compounds called salicylates. Aspirin
use as medicine like analgesic
(pain-relieving), antipyretic (fever-reducing),
and anti-inflammatory but now days it is commonly used as an antiplateletagent in the treatment and prevention of heart attacks and strokes. Aspirin is a
synthetic compound.
Like
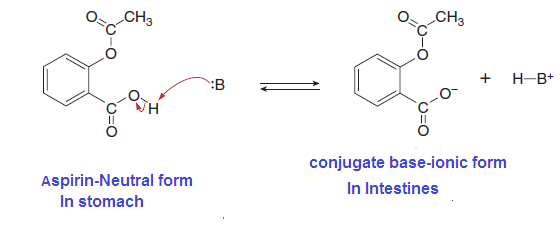
many drugs, aspirin is capable of undergoing a proton transfer reaction. Its
most acidic proton is the H bonded to O, and in the presence of base, this H is
readily removed.
Acetylsalicylic
acid does not occur in nature, though some related salicylates are found in
willow bark and meadowsweet blossoms.
Similar Compounds:
Similar Compounds:

No comments:
Post a Comment