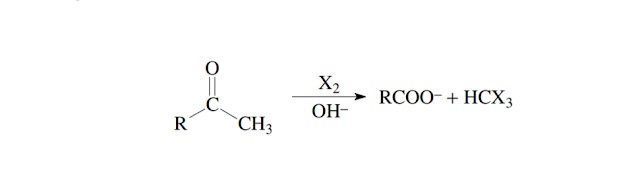
The oxidative cleavage
of Methyl ketones as well as acetaldehyde and 2° terminal alcohol into into a
carboxylate anion and a trihalomethane (haloform) by the reaction of one
of halogen in the presence of strong alkali like NaOH or KOH is known as Haloform
reaction.
The respective halogen
can be chlorine, bromine or iodine.The methyl group of a methyl ketone is converted
into an trihalomethane by multiple subsequent steps, that involve
formation of an intermediate enolate anion.
Related Questions:
Chlorination of ethane to ethyl chloride is more practicable than chlorination of n-Pentane to 1-Chloropentanc. Why ?
No comments:
Post a Comment