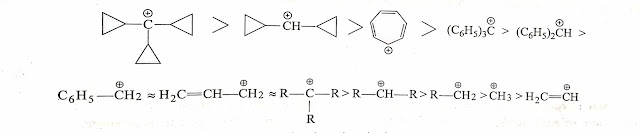
Actually answers of this question is always confusing, most of the authors believe benzyl carbocation is more stable than tertiary because benzyl carbocation involves in resonance.
But some of the authors believe that Tertiary carbocation is more stable as it involves maximum +I effect and maximum hyperconjuation +H (9-alpha hydrogens). Maximum +I and +H is more dominant than +M effect. Thus tertiary carbocation is more stable than benzyl carbocation.
Important note:
Stability of Benzyl , allylic and tertiary alkyl carbocation is practically almost same .so that stabilities infact cannot be compared.
Similar Questions:
good post
ReplyDeleteThank you for comments
DeleteThanks, you saved my ass
ReplyDeleteHow
DeleteI have a doubt in that stability order because my teacher teach me another stability order it seems to be little bit different to seeing the pictures I can post my teachers teached stability order if any will tell it is right or wrong
ReplyDelete