Basicity of Guanidine:
Guanidine is the strongest base among neutral compounds:
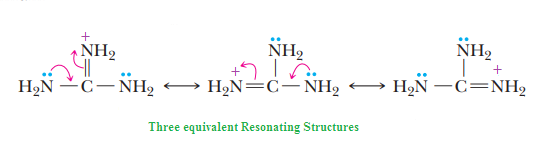
The remarkable basicity of guanidine is attributed to the fact that the positive charge on the guanidinium ion is delocalized equally over the three nitrogen atoms, as shown by
these three equivalent resonating structures:
Basicity of nitrogen can be increased by attachment topi-donors (NH2) group. These two pi-donating NH2groups donate electron density to the (pi-accepting) C=NH.

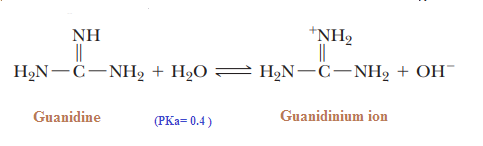
No comments:
Post a Comment