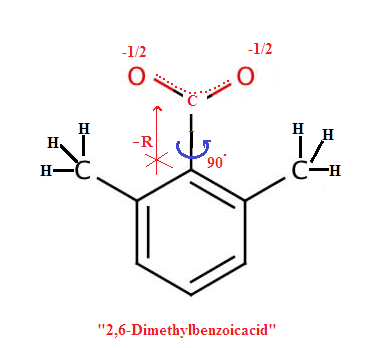
Ortho substituted Benzoic acid is more acidic then meta or para substituted benzoic acid irrespective of nature of group (electron donating group or electron withdrawing group)
When a group present in the ortho position with respect to carboxylic group creates steric strain resulting in rotation of the carboxylic group and shifting it out of plane of the benzene ring as a resultthe carboxylic group can no longer participate in ring resonance and thereby the acidity increases as delocalization of negative charge equally on the both oxygen atom of conjugate base ( carboxylate ion) of the benzoic acid , hence carboxylate ion more stablised. This is also called Ortho effect.
Note: However for groups like -NH2 or -OH does not experience SIR effect due to small size and –CN is linear group so SIR effect not applicable.

No comments:
Post a Comment