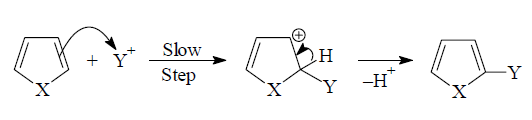
Pyrrole, furan and thiophene undergo electrophilic substitution
reactions like nitration, sulphonation, halogenation etc. characteristic of
aromatic rings. That carbons in 5-membered heterocyclic rings have higher
electron density compared to benzene and hence undergo electrophilic
substitution more readily than benzene.The electrophilic substitution takes
place preferentially at 2-position (C-2).
The attack of an electrophile on pyrrole, for
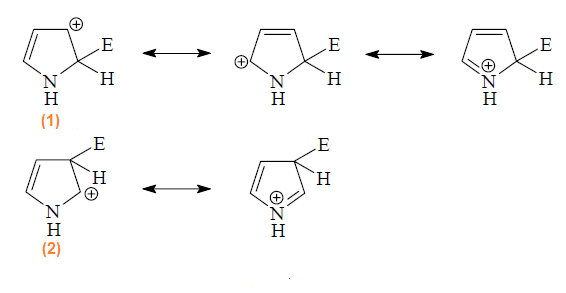
example, will lead to formation of 2- and 3-substitution products by way of
carbocations (1) and (2) respectively.The substitution occurs preferably at C-2 position
because the intermediate obtained by attack at this position is more stable
than the intermediate obtained by attack at C-3. The positive charge in
intermediate (1) is more delocalized than
intermediate (2) and hence is more stable
and preferred intermediate.
The electrophilic
substitution at C-2 in furan and thiophene can also be accounted in the same
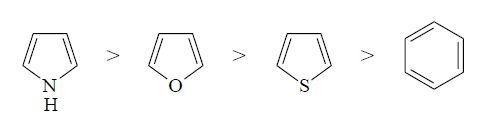
manner. Furan is not as reactive as pyrrole in electrophilic substitution
reactions because the oxygen in furan is more electronegative than nitrogen in
pyrrole and therefore does not enhance the electron density of carbons as much
as pyrrole. Thiophene is less reactive than furan towards electrophilic
substitution because the p-electrons of sulphur are in 3p orbital which
overlaps less effectively than the 2p orbital of nitrogen or oxygen with 2p
orbitals of carbon. The relative reactivities towards electrophilic
substitution follows the order:
SIR effect:
Dancing Resonance:
Inversion of Amines: