Related Questions:
Search This Blog
Saturday, May 30, 2020
Why are bridge head carbocations unstable?
According to Bredt’s rule a bridgehead carbon atom of bicyclo compound cannot be sp2 hybridised or in other word a bridgehead carbon atom cannot be form double bond. unless the ring that contains at least eight atoms.
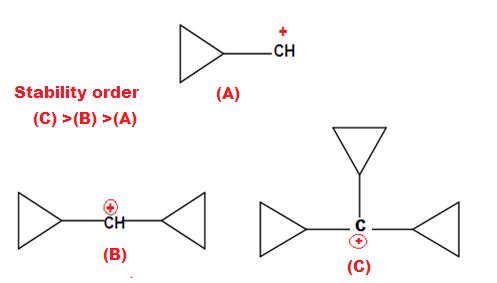
Why is cyclopropyl methyl carbocation exceptionally stable?
The exceptional stability of cyclopropane methyl cation can be explained by the concept of dancing resonance concept. The stability of additional cyclopropyl group , is result of more conjugation between the bent orbital of cyclopropyl ring and cationic carbon.
The most stable carbocation known till date in organic chemistry is explain by Dancing resonance.
Related Questions:
Friday, May 29, 2020
Azabicyclo[2,2,1]heptane is more basic thantriethylamine why?.
Subscribe to:
Comments (Atom)