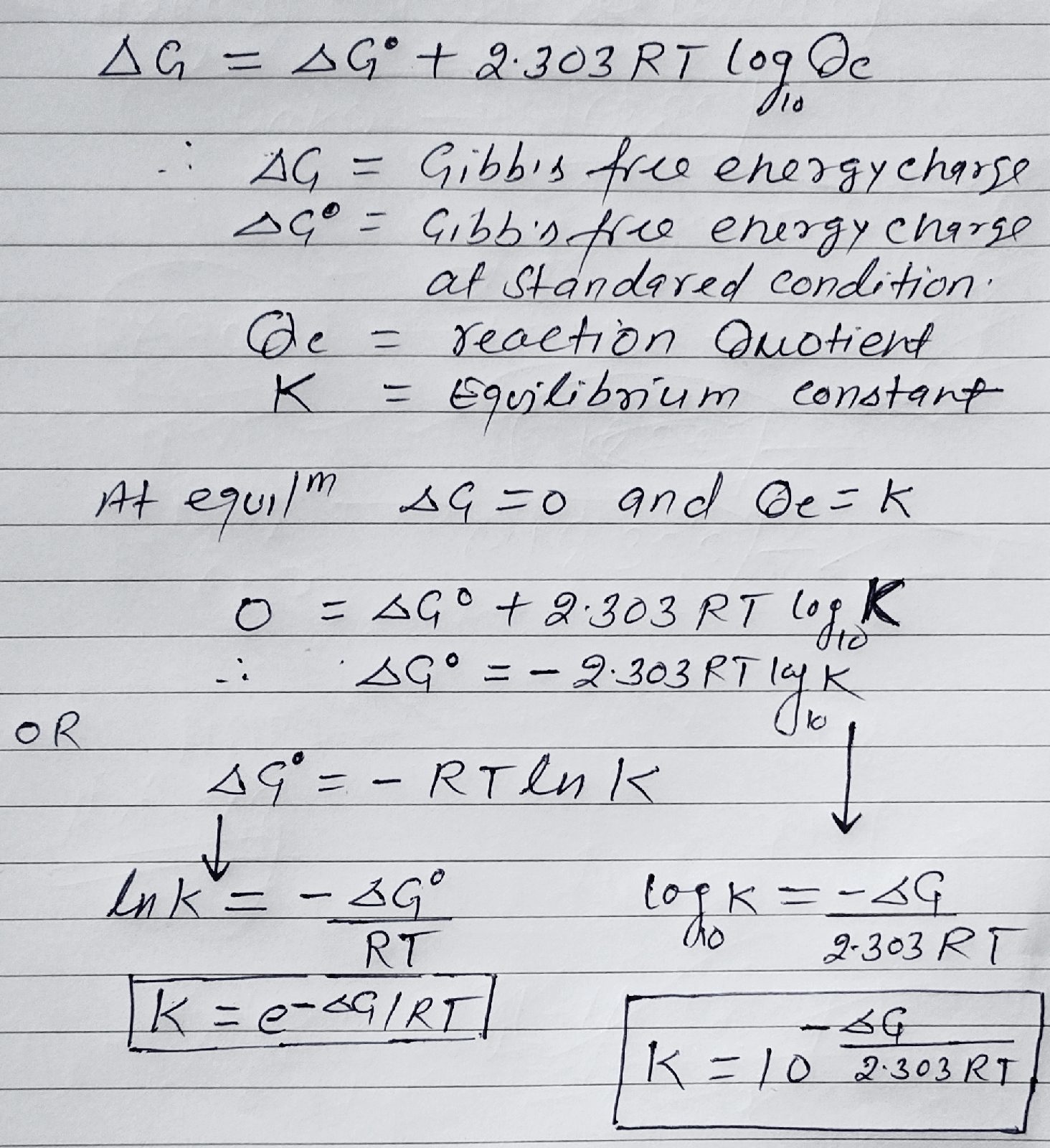Search This Blog
Tuesday, September 5, 2023
The rate constant for zero order reaction is 2× 10^-2 Mole L^-1 sec-1. If the concentration of reactant after 25 sec is 0.5M ,the initial concentration must have been ?
(A) 1M (B) 2M (C) 1.5M (D) 2.5M
Consider a general reaction A--->B follow zero order kinetics.if concentration of A is reduces from 0.1 M to 0.05 M in 10 second then calculate time taken when concentration reduce to 0.025 M.
(A) 2.5 sec (B) 2 sec (C) 5 sec (D) 10 sec
Sunday, September 3, 2023
Equilibrium constant for the reaction given below is 2.0 × 10^-7 at 300 K. calculate standard Gibb's free energy change for the reaction PCl5 (g) --=--PCl3 (g) + Cl2 (g) also calculate standard entropy change if ∆H° = 28.40 Kj mole-1.
Saturday, September 2, 2023
Gibb's free energy (∆G) and Equilibrium constant (Kc):
The part f energy which is converted into usefull work called Gibb's free energy or Gibb's function.
Energy (H) = Useful work (G) + Non useful (TS)
We van not calculate absolute value of 'G' so we calculate change in Gibb's free energy.
∆G = ∆H -∆TS
∆G = ∆H - (∆TS + T∆S). ......(1)
Standard Gibb's energy (∆G°) change at standard condition is 1 bar and 298 K
∆G° = ∆H° -∆TS° .......(2)
Relation between ∆G° and Equilibrium constant (K):
Topic:
CHEMICAL EQUILIBRIUM:
Subscribe to:
Comments (Atom)