We know that on increasing the entropy of a pure crystalline
substance increases because molecular motion increases with increase of
temperature and decreases on decreasing temperature. Or we can say that the entropy of a perfectly crystalline
solid approaches zero as the absolute zero of temperature is approached. Which
means that at absolute zero every crystalline solid is in a state of perfect
order and its entropy should be zero. This
is third law of thermodynamics.
We can calculate absolute value of entropy of any
substance in any state (solid ,liquid, gas) at any temperature by calculating
dS for the processes in going from the initial state to the state of the
substance for which entropy is to be calculated.
EXAMPLE: Find the entropy of change when 90 g
of H2O at 10C was converted into steam at 100C.(
Given Cp(H2O)=75.29 JK-1 and dHVap=43.932 k JK-1 mol-1)
SOLUTION:
EXAMPLE: Calculate dG for
(i) H2O
(l, 1 atm, 300 K ) ------à H2O (g, 1
atm, 300 K)
SOLUTION: (1)
(2)





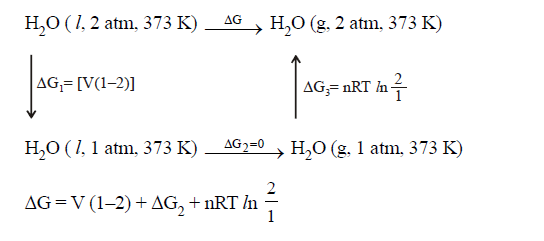
No comments:
Post a Comment