The salient features of
fluorite structure are:
(1) The Ca+2 ions are arranged in ccp arrangement, i.e. these ions
occupy all the corners and the centres of each face of the cube
(2) The F– ions occupy all the tetrahedral holes.
(3) Since there are two tetrahedral holes for each Ca+2 ion
and F- ions occupy all the tetrahedral holes, there will be two F- ions for each Ca+2
ions, thus the stoichiometry of the compound is 1:2.
(4) Each Ca+2
ion is surrounded by 8F- ions and each F- ions is surrounded by 4Ca+2
ions. The Coordination number of Ca+2 ion is eight and
that of F- ion is four, this is called 8:4 Coordination.
(5) Each
unit cell has 4 calcium ions and 8 fluoride ions so formula of unit cell is Ca4F8
which is explained as below
No.
of Ca+2 ions = 8(at corners)´1/8 + 6 (at face centres)´1/2
No.
of F ions = 8 (within the body)´1 = 8
Thus the number of CaF2 units per unit cell is 4.
(6) Radius Ratio, packing efficiency ,void % and
density:
Other
examples: of structure are SrF2, BaCl2, BaF2,
PbF2, CdF2, HgF2, CuF2, SrCl2,
etc.


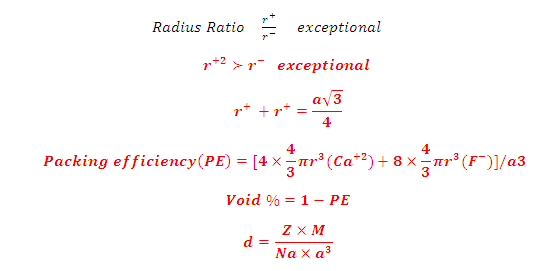
No comments:
Post a Comment