VAPOUR PRESSURE:
(1) If a sample of water in its liquid
phase is placed in an empty container, some of it will vaporize to form gaseous
of water. This change is called evaporation.
(2) The pressure exerted by the vapour (molecules
in the vapour phase) over the surface of the liquid at the equilibrium at given
temperature is called the vapour pressure of the liquid.
OR
(3)
It is the pressure exerted by the vapour when vapours are equilibrium with the
liquid.
(4)
The pressure exerted by vapours is called unsaturated vapour pressure or partial vapour at
non equilibrium condition.
Factors affecting vapour
pressure:
(A) Temperature:.
(1) The temperature at which the vapour
pressure of the liquid becomes equal to the atmospheric pressure is called its boiling point.
(2) Vapour
pressure is directly
proportional to the Temperature so that on increasing
temperature the rate of evaporation increases and rate of condensation
decreases and hence vapour pressure increases.
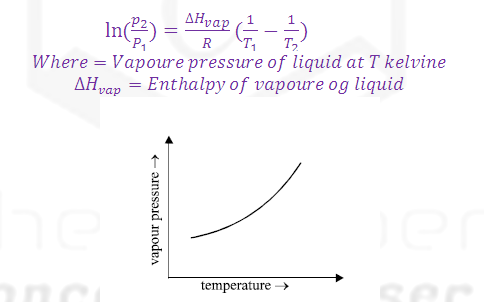
(3) The dependence of vapour pressure and
temperature is given by CLASIUS CLAPERON equation.
(4) Vapour pressure of a particular
liquid system is only the function of temperature only. It is independent from
all other factors like surface area, amount of liquid, available space etc.
(A) Nature of liquid:
Vapour
pressure of liquid =1/the strength of intermolecular forces acting
between molecules
For example: CCl4
has higher vapour pressure because of the weak intermolecular forces acting
between its molecules than water which has stronger intermolecular forces
acting between water molecules of volatile liquid has lower boiling point than
a non-volatile liquid.
Note:
(1)
Relative lowering of vapour pressure of a solvent is a colligative property
equal to the vapour pressure of the pure solvent minus the vapour pressure of
the solution.
(2) For example: water at 20°C
has a vapour pressure of 17.54 mmHg. Ethylene glycol is a liquid whose vapour
pressure at 20°C is relatively low, an aqueous solution containing
0.010 mole fraction of ethylene glycol has a vapour pressure of 17.36 mmHg.
Thus the vapour pressure lowering, DP
= 17.54 mmHg ¾ 17.36 mmHg = 0.18 mmHg.
RAOULT’S LAW:
(1)
Vapour pressure of a number of binary solutions of volatile liquids such as
benzene and toluene at constant temperature gave the following generalization
which is known as the Raoult’s law.
(2) Raoult’s law states that “The
partial pressure of any volatile component of a solution at any temperature is
equal to the vapour pressure of the
pure component multiplied by the mole
fraction of that component in the solution
(A) Vapour pressure of liquid-liquid
Solution:
(3)
Suppose a binary solution contains nA moles of a volatile liquid A and nB
moles of a volatile liquid B, if PA and PB are partial
pressure of the two liquid components, the according to Raoult’s law
(4) If the vapour behaves like an ideal
gas, then according to Dalton’s law
of partial pressures, the total pressure P is given by
Graphical representation of Raoult’s law:
(5) The relationship between vapour pressure and mole fraction of an ideal
solution at constant temperature is shown. The dashed lines 1 and 2 represent
the partial pressure of the components. The total vapour pressure is given by 3rd
line in the above figure.
(B) Vapour pressure of Solid-liquid
Solution:
(1)
Vapor pressure, when a small amount of a non-volatile solute (solid) is added
to the liquid (solvent). It is found that the vapour pressure of the solution
is less than that of the pure solvent.
(2)
The lowering of vapour pressure is due to the fact that the solute particles
occupy a certain surface area and evaporation takes place from the surface
only. and
(3)
The particles of the solvent will have a less tendency to change into vapour
i.e. the vapour pressure of the solution will be less than that of the pure
solvent and it is termed as lowering of vapour pressure.
For a solution of
non-volatile solute with volatile solvent.
ILLUSTRATIVE EXAMPLE (1): The vapour pressure of ethanol and methanol are
44.5 mm and 88.7 mm Hg respectively. An ideal solution is formed at the same
temperature by mixing 60 g of ethanol with 40g of methanol. Calculate total
vapour pressure of the solution.
SOLUTION:
ILLUSTRATIVE EXAMPLE (2): What is the composition of the vapour which is in
equilibrium at 30°C with a benzene-toluene solution with a mole fraction of
benzene of 0.400?
SOLUTION:
ILLUSTRATIVE EXAMPLE (3): The
composition of vapour over a binary ideal solution is determined by the
composition of the liquid. If XA and
YA are the mole-fraction of A in the liquid and vapour, respectively
find the value of XA for which YA-XA has a minimum. What is the value of the
pressure at this composition?
SOLUTION
ILLUSTRATIVE EXAMPLE (4): One mole of
a non-volatile solute is dissolved in two moles of water. The vapour pressure
of the solution relative to that of water is
SOLUTION:
Mole fraction of solute in
solution
Raoult's Law v/s Dalton's Law: Determination of composition in vapour phase: Coming soon..











No comments:
Post a Comment