According to bent rule more electronegative atom or group attached those orbital have minimum S- character. There is in Trigonal bipiramidal (TBP) Geometry we known that axial orbital hare no S- character so F atom attached with axial positions only. Hence PCl3F2 has zero dipole moment.
Search This Blog
Sunday, May 24, 2020
What is Banana bond (3C-2e bridge bond) ? Explaine with suitable examples.
3C-2e BOND OR BANANA BOND:
EXAMPLE FORMATION OF B2H6:
(1) Formation of 3C-2e bond in B2H6 is best explain by MOT and total number of bond in B2H6 is 6 (3C-2e=2 and 3C-4e=4)
(2) Bridge bonds are longer than terminal bond because at bridge bonds electrons are delocalized at three centres
(3) Bond energy (441kj/mole) of B-H-B bond is greater than bond energy (381 K j/mole) of B-H bond.
(4) Hybridization of B atom is sp3, so non planer, and non polar (U=0)
(5) B2H6 Methylated up to B2H2 Me4
(6) B2H6 is hypovalent molecule hence act as Lewis acid and undergoes two type of cleavage when react with Lewis base
What is bridge bond ? explaine 3C-4e bridge bond with suitable examples .
3C-4e BOND or 3C-4e BRIDGE BOND:
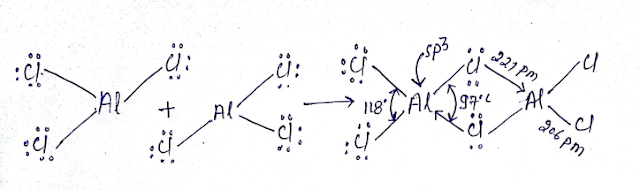
Al2Cl6 Dimmerised by 3C-4e bond bridge bond:
Al2Cl6 is neither hypovalent nor hypovalent rather its octet is complete. We will used MOT here it cannot act as Lewis acid due to crowding in spite having vacant d orbital’s however Alcl3 act as Lewis acid.
Al2Cl6 contains six bond having two bridge bond(3c-4e) and four bond is (2C-2e)
Boron do not formed bridge bond because boron experience steric crowding.
Topic:
Bridge Bonding,
CHEMICAL BONDING:
Subscribe to:
Comments (Atom)