Search This Blog
Wednesday, May 29, 2024
Titration of NaOH ( Titrate ) with CH3COOH (Titrant)
Sunday, May 26, 2024
Derivation of Van't Hoff equation.
Wednesday, January 10, 2024
Why Bond length of O-O is greater in H2O2 than O2F2?
Electronegativity of F is much more than hydrogen and also hybridization of oxygen atoms in of H2O2 and O2F2 both have (sp3) same and hence we can apply bent’s rule.
According bent’s rule more eletronegative atoms reduce % s-character (or increases % p-character vice versa) of those hybride orbital in which they attach. So in case of O2F2 % s-character of those hybride orbital decrease which have flourine while % p-character increases in same way % s-character of remaing hybride orbital inceases and p-character decreases hence its bond length also (bond length is directly proportional to % p-character) decreases. hence O-O bond length in O2F2 is shorter thane H2O2.
Wednesday, October 18, 2023
“तलाश” मेरी क़लम से …
गुजरी है करीब से कई बहारे , किसी की महक से ये वजूद महका ही नहीँ ,
न जाने कहाँ है कौन है ओ । जिसकी तलाश मे दर बदर मै भटकता हूँ ,
कही तो ये तलाश ख़त्म होगीं , इस सफ़र की ,जो मंजिलें कहीँ खो गयी है ,
कभी तो मिलेंगी इसी इंतज़ार मे , चिलमन गिराये बैठा हूँ ॥ मै तेरी तलाश मे , हाँ तेरी तलाश मे , हाँ तेरी तलाश मे ॥
(Written by: सुनील कुमार "सकल")
Sunday, October 15, 2023
The activation energy of a reaction is 225 k Cal mol-1 and the value of rate constant at 40°C is 1.8 ×10^−5 s^−1 . Calculate the frequency factor, A.
SOLUTION:
Here, we are given that
Ea = 22.5 kcal mol-1 = 22500 cal mol-1
T = 40°C = 40 + 273 = 313 K
k = 1.8 × 10-5 sec-1
Substituting the values in the equation
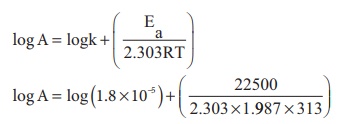
log A = log (1.8) −5 + (15.7089)
log A = 10.9642
A = antilog (10.9642)
A = 9.208 × 101^0 collisions s^−1



