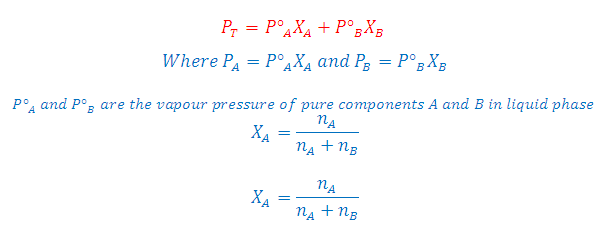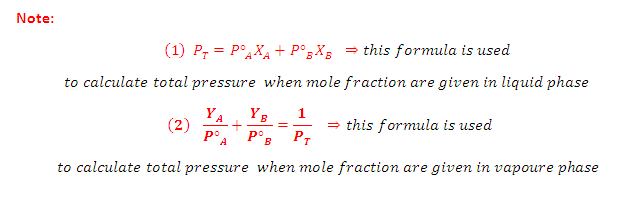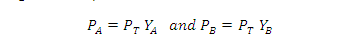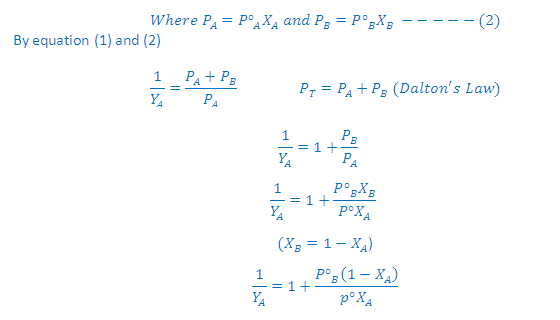In order to understand the composition
of the compounds, it is necessary to have a theory which accounts for both
qualitative and quantitative observations during chemical changes. Observations
of chemical reactions were most significant in the development of a
satisfactory theory of the nature of matter. These observations of chemical
reactions are summarized in certain statements known as laws of chemical
combination.
Search This Blog
Sunday, May 12, 2019
Wednesday, May 1, 2019
SOLUBILITY OF GASES AND HENRY’S LAW:
Solubility:
Solubility of a substance is its maximum
amount that can be dissolved in a specified amount of solvent at a specific
temperature. It depends upon the nature of solute and solvent as well as
temperature and pressure.
Unit:
Unit of Solubility is gm/litre
or Mole/Litre
(A) Solubility of Solid in a Liquid:
Every solid does not dissolve in a given liquid. While sodium chloride and sugar dissolve readily in water, naphthalene and anthracene do not. On the other hand, naphthalene and anthracene dissolve readily in benzene but sodium chloride and sugar do not.
Every solid does not dissolve in a given liquid. While sodium chloride and sugar dissolve readily in water, naphthalene and anthracene do not. On the other hand, naphthalene and anthracene dissolve readily in benzene but sodium chloride and sugar do not.
(1)
It is clear observed that polar solutes dissolve in polar
solvents and non polar solutes in non-polar
solvents.
(2)
In general, a solute dissolves in a solvent if the intermolecular interactions
are similar in the two or we may say like dissolves like.
(3) Dissolution: When a solid solute is added to the solvent, some
solute dissolves and its concentration increases in solution. This process is
known as dissolution.
(4) Crystallisation: Some solute particles in solution collide with the
solid solute particles and get separated out of solution. This process is known
as crystallisation.
“A stage is
reached when the two processes (dissolution and crystallisation) occur at the same rate. Under such
conditions, number of solute particles going into solution will be equal to the
solute particles separating out and a state of dynamic equilibrium is reached.
At this stage the concentration of solute in solution will remain constant
under the given conditions, i.e., temperature and pressure. Similar process is
followed when gases are dissolved in liquid solvents.
(5) Saturated solution: Such a solution in which no more
solute can be dissolved at the same temperature and pressure is called a saturated solution.
(6) Unsaturated solution: The Solution in which more solute can be
dissolved at the same temperature.
(7) The solution which is in dynamic equilibrium with
undissolved solute is the saturated solution and contains the maximum amount of
solute dissolved in a given amount of solvent. Thus, the concentration of
solute in such a solution is its solubility.
Factors affecting
Solubility:
Earlier we have observed that solubility of one substance into another depends on the nature of the substances. In addition to these variables, two other parameters, i.e., temperature and pressure also control this phenomenon.
(1) Effect of temperature:
The solubility of a solid in a liquid is significantly affected by temperature changes. Consider the equilibrium exist between dissolution and crystallisation. This, being dynamic equilibrium, must follow Le Chateliers Principle. In general…….
Earlier we have observed that solubility of one substance into another depends on the nature of the substances. In addition to these variables, two other parameters, i.e., temperature and pressure also control this phenomenon.
(1) Effect of temperature:
The solubility of a solid in a liquid is significantly affected by temperature changes. Consider the equilibrium exist between dissolution and crystallisation. This, being dynamic equilibrium, must follow Le Chateliers Principle. In general…….
(i) If in a nearly saturated solution,
the dissolution process is endothermic (Δsol H > 0), the solubility
should increase with rise in temperature and
(ii) If it is exothermic (Δsol H > 0) the
solubility should decrease. These trends are also observed experimentally.
(2) Effect of Pressure:
Pressure does not have any significant effect on solubility of solids in liquids. It is so because solids and liquids are highly incompressible and practically remain unaffected by changes in pressure.
(2) Effect of Pressure:
Pressure does not have any significant effect on solubility of solids in liquids. It is so because solids and liquids are highly incompressible and practically remain unaffected by changes in pressure.
(2) Solubility of gas in
Liquid:
Many gases dissolve in water. Oxygen dissolves only to a small extent in water. It is this dissolved oxygen which sustains all aquatic life. On the other hand, hydrogen chloride gas (HCl) is highly soluble in water. Solubility of gases in liquids is greatly affected by pressure and
temperature.
Many gases dissolve in water. Oxygen dissolves only to a small extent in water. It is this dissolved oxygen which sustains all aquatic life. On the other hand, hydrogen chloride gas (HCl) is highly soluble in water. Solubility of gases in liquids is greatly affected by pressure and
temperature.
Factors affecting
Solubility:
(1) Effect of Pressure:
The solubility of gases increase with
increase of pressure. For solution of gases in a solvent, consider a solution
is act as system and that system to be in a state of dynamic equilibrium,
i.e., under these conditions rate of gaseous particles entering and leaving the
solution phase is the same. Now increase the pressure over the solution phase
by compressing the gas to a smaller volume, this will increase the number of gaseous
particles per unit volume over the solution and also the rate at which the
gaseous particles are striking the surface of solution to enter it. The
solubility of the gas will increase until a new equilibrium is reached
resulting in an increase in the pressure of a gas above the solution and thus
its solubility increases.
Henry’s Law:
(1) The solubility of a gas in a liquid
is determined by several factors. In addition to the nature of the gas and the
liquid, solubility of the gas depends on the temperature and pressure of the
system.
(2) The
solubility of a gas in a liquid is governed by Henry’s law which states that the
solubility of a gas in a liquid is directly proportional to the pressure of the
gas.
(3) Dalton, a contemporary of Henry, also
concluded independently that the solubility of a gas in a liquid solution is a
function of the partial pressure of the gas. If we use the mole fraction of the
gas in the solution as a measure of its solubility, then: Mole fraction of the
gas in a solution is proportional to the partial pressure of the gas.
Or, partial pressure of the gas in solution = KH ´ mole fraction of the gas in solution
Here KH is Henry’s law constant
p = KH X
(Solute)
If we draw a graph between partial pressure of
the gas versus mole fraction of the gas in solution, then we should get a plot
of the straight line passing through origin.
Experimental result for the solubility of HCl gas in Cyclohexane
at 93 K the slope of line is the Henry’s law constant
Different gases have different KH
values at the same temperature. This suggests that KH is a function
of the nature of the gas. Table gives KH values of some common gases
at specified temperature
Values of Henry’s law constant (KH) for some selected
gases in water:
It is obvious from figure that the
higher the value of KH at a given pressure, the lower is the
solubility of the gas in the liquid. It can be seen from table that KH value for both N2 and O2 increases
with increase in temperature indicating that solubility of gases decreases with
increase of temperature. It is due to this reason that aquatic
species are more comfortable in cold waters rather than warm waters.
Application of Henry’s Law:
Henry’s law finds several applications in industry and explains some biological phenomena Notable among these are:
Henry’s law finds several applications in industry and explains some biological phenomena Notable among these are:
(1) To increase the solubility of CO2 in
soft drinks and soda water, the bottle is sealed under high pressure.
(2) To minimize the painful effects accompanying
the decompression of deep sea divers, oxygen diluted with less soluble helium
gas is used as breathing gas.
(3) In lungs, where oxygen is present in air with
high partial pressure, haemoglobin combines with oxygen to form oxyhaemoglobin.
In tissues where partial pressure of oxygen is low, oxyhaemoglobin releases oxygen
for utilization in cellular activities.
(1) At High Pressure:
Scuba divers must cope with high concentrations
of dissolved gases while breathing air at high pressure underwater. Increased
pressure increases the solubility of atmospheric gases in blood. When the divers
come towards surface, the pressure gradually decreases. This releases the
dissolved gases and leads to the formation of bubbles of nitrogen in the blood.
This blocks capillaries and creates a medical condition
known as bends, which are painful and dangerous to life.
To avoid bends, as well as, the toxic effects
of high concentrations of nitrogen in the blood, the tanks used by scuba divers
are filled with air diluted with helium (11.7% helium, 56.2% nitrogen and 32.1% oxygen).
(2) At Low Pressure: At high altitudes the partial pressure of oxygen is less than that at the ground level. This leads to low concentrations of oxygen in the blood and tissues of people living at high altitudes or climbers. Low blood oxygen causes climbers to become weak and unable to think clearly, symptoms of a condition known as anoxia.
(2) At Low Pressure: At high altitudes the partial pressure of oxygen is less than that at the ground level. This leads to low concentrations of oxygen in the blood and tissues of people living at high altitudes or climbers. Low blood oxygen causes climbers to become weak and unable to think clearly, symptoms of a condition known as anoxia.
(2) Effect of
temperature:
Solubility of gases in liquids decreases with rise in temperature. When dissolved, the gas molecules are present in liquid phase and the process of dissolution can be considered similar to condensation and heat is evolved in this process. We have known that dissolution process involves dynamic equilibrium and thus must follow Le Chatelier’s Principle. As dissolution is an exothermic process, the solubility should decrease with increase of temperature.
Solubility of gases in liquids decreases with rise in temperature. When dissolved, the gas molecules are present in liquid phase and the process of dissolution can be considered similar to condensation and heat is evolved in this process. We have known that dissolution process involves dynamic equilibrium and thus must follow Le Chatelier’s Principle. As dissolution is an exothermic process, the solubility should decrease with increase of temperature.
ILLUSTRATIVE EXAMPLE (1): If N2
gas is bubbled through water at 293 K, how many millimoles of N2 gas
would dissolve in 1 litre of water. Assume that N2 exerts a partial
pressure of 0.987 bar. Given that Henry’s law constant for N2 at 293
K is 76.84 kbar.
SOLUTION: The
solubility of gas is related to its mole fraction in the aqueous solution. The
mole fraction of the gas in the solution is calculated by applying Henry’s law.
Thus,
As 1litre water contains 55.5 mol of it, therefore, if n represents number of moles of N2 in solution,
Tuesday, April 30, 2019
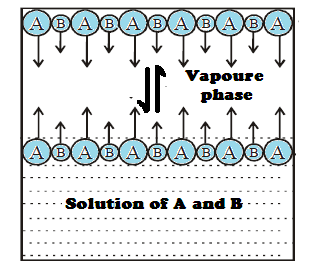
DALTON'S LAW VERSES RAOULT'S LAW:
Determination of composition
in vapour phase:
The composition of the vapour in equilibrium with
the solution can be calculated applying Daltons’ law of partial pressures. Let
the mole fractions of vapours A and B be YA and YB respectively. Let PA
and PB
be the partial pressure of vapours A and B respectively and total pressure PT.
In Vapours phase:
YA=
mole fraction of A in vapour phase
YB
= mole fraction of B in vapour phase
(YA+YB =1)
In liquid solution phase:
XA
= mole fraction of A in liquid phase
XB
= mole fraction of B in liquid phase
(XA + XB = 1)
According to Raoult’s Law: The partial
pressure of any volatile component of a solution at any temperature is equal to
the vapour pressure of the pure component multiplied by the mole fraction of
that component in the solution.
Where
XA and XB is the mole fraction of the component A and B in
liquid phase respectively
According to Dalton’s Law:
The vapour behaves like an ideal gas, then according to Dalton’s law of partial
pressures, the total pressure PT is given by:
Partial
pressure of the gas = Total pressure x Mole fraction
PA = PT YA
and PB =PT YB
Where YA and YB is the
mole fraction of the component A and B in gas phase respectively
Combination of Raoult’s and Dalton’s Law:
(3) Thus, in case of ideal solution the vapour
phase is phase is richer with more volatile component i.e., the one having
relatively greater vapour pressure
Graph Between 1/YA Vs 1/XA:
According
to Dalton’s law of partial pressures, the total
pressure PT is given by:
Partial
pressure of the gas = Total pressure x Mole fraction
Where YA and YB is the
mole fraction of the component A and B in gas phase respectively
According
to Raoult’s law:
On rearrangement of this
equation we get a straight line equation:
Monday, April 29, 2019
VAPOUR PRESSURE AND RAOULT’S LAW:
VAPOUR PRESSURE:
(1) If a sample of water in its liquid
phase is placed in an empty container, some of it will vaporize to form gaseous
of water. This change is called evaporation.
(2) The pressure exerted by the vapour (molecules
in the vapour phase) over the surface of the liquid at the equilibrium at given
temperature is called the vapour pressure of the liquid.
OR
(3)
It is the pressure exerted by the vapour when vapours are equilibrium with the
liquid.
(4)
The pressure exerted by vapours is called unsaturated vapour pressure or partial vapour at
non equilibrium condition.
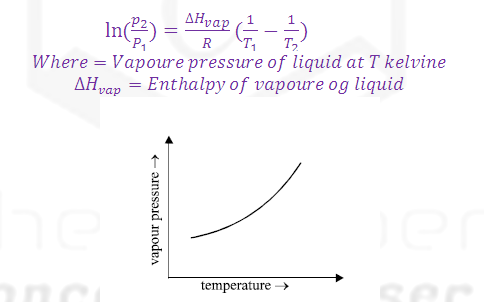
Factors affecting vapour
pressure:
(A) Temperature:.
(1) The temperature at which the vapour
pressure of the liquid becomes equal to the atmospheric pressure is called its boiling point.
(2) Vapour
pressure is directly
proportional to the Temperature so that on increasing
temperature the rate of evaporation increases and rate of condensation
decreases and hence vapour pressure increases.
(3) The dependence of vapour pressure and
temperature is given by CLASIUS CLAPERON equation.
(4) Vapour pressure of a particular
liquid system is only the function of temperature only. It is independent from
all other factors like surface area, amount of liquid, available space etc.
(A) Nature of liquid:
Vapour
pressure of liquid =1/the strength of intermolecular forces acting
between molecules
For example: CCl4
has higher vapour pressure because of the weak intermolecular forces acting
between its molecules than water which has stronger intermolecular forces
acting between water molecules of volatile liquid has lower boiling point than
a non-volatile liquid.
Note:
(1)
Relative lowering of vapour pressure of a solvent is a colligative property
equal to the vapour pressure of the pure solvent minus the vapour pressure of
the solution.
(2) For example: water at 20°C
has a vapour pressure of 17.54 mmHg. Ethylene glycol is a liquid whose vapour
pressure at 20°C is relatively low, an aqueous solution containing
0.010 mole fraction of ethylene glycol has a vapour pressure of 17.36 mmHg.
Thus the vapour pressure lowering, DP
= 17.54 mmHg ¾ 17.36 mmHg = 0.18 mmHg.
RAOULT’S LAW:
(1)
Vapour pressure of a number of binary solutions of volatile liquids such as
benzene and toluene at constant temperature gave the following generalization
which is known as the Raoult’s law.
(2) Raoult’s law states that “The
partial pressure of any volatile component of a solution at any temperature is
equal to the vapour pressure of the
pure component multiplied by the mole
fraction of that component in the solution
(A) Vapour pressure of liquid-liquid
Solution:
(3)
Suppose a binary solution contains nA moles of a volatile liquid A and nB
moles of a volatile liquid B, if PA and PB are partial
pressure of the two liquid components, the according to Raoult’s law
(4) If the vapour behaves like an ideal
gas, then according to Dalton’s law
of partial pressures, the total pressure P is given by
Graphical representation of Raoult’s law:
(5) The relationship between vapour pressure and mole fraction of an ideal
solution at constant temperature is shown. The dashed lines 1 and 2 represent
the partial pressure of the components. The total vapour pressure is given by 3rd
line in the above figure.
(B) Vapour pressure of Solid-liquid
Solution:
(1)
Vapor pressure, when a small amount of a non-volatile solute (solid) is added
to the liquid (solvent). It is found that the vapour pressure of the solution
is less than that of the pure solvent.
(2)
The lowering of vapour pressure is due to the fact that the solute particles
occupy a certain surface area and evaporation takes place from the surface
only. and
(3)
The particles of the solvent will have a less tendency to change into vapour
i.e. the vapour pressure of the solution will be less than that of the pure
solvent and it is termed as lowering of vapour pressure.
For a solution of
non-volatile solute with volatile solvent.
ILLUSTRATIVE EXAMPLE (1): The vapour pressure of ethanol and methanol are
44.5 mm and 88.7 mm Hg respectively. An ideal solution is formed at the same
temperature by mixing 60 g of ethanol with 40g of methanol. Calculate total
vapour pressure of the solution.
SOLUTION:
ILLUSTRATIVE EXAMPLE (2): What is the composition of the vapour which is in
equilibrium at 30°C with a benzene-toluene solution with a mole fraction of
benzene of 0.400?
SOLUTION:
ILLUSTRATIVE EXAMPLE (3): The
composition of vapour over a binary ideal solution is determined by the
composition of the liquid. If XA and
YA are the mole-fraction of A in the liquid and vapour, respectively
find the value of XA for which YA-XA has a minimum. What is the value of the
pressure at this composition?
SOLUTION
ILLUSTRATIVE EXAMPLE (4): One mole of
a non-volatile solute is dissolved in two moles of water. The vapour pressure
of the solution relative to that of water is
SOLUTION:
Mole fraction of solute in
solution
Raoult's Law v/s Dalton's Law: Determination of composition in vapour phase: Coming soon..
Sunday, April 28, 2019
IDEAL AND NON IDEAL SOLUTIONS:
An
ideal solution of substance A and B is one in which both substances follow
Raoult’s law for all values of mole fractions. Such solutions occurs when the
substances are chemically similar so that the intermolecular forces between A
and B molecules are similar to those between two A molecules or between two B
molecules. The total vapour pressure over an ideal solution equals the sum of
the partial vapour pressures, each of which is given by Raoult’s law.
(1) Condition for a solution
to be Ideal
Two liquids on mixing form an ideal solution
only when
(1) Both have similar structures and polarity
so that they have similar molecular environment.
(2) Both have similar molecular sizes.
(3) Both have identical intermolecular forces.
(4) The liquid should not dissociate or
associate each other.
(5)
For the solid solute, solution must be extremely diluted.
(2) Characteristic of Ideal Solution:
(3) Examples of Ideal
Solution: Solution of benzene C6H6
and toluene C6H5CH3 are ideal. Note the
similarity in their structural formula.
Suppose a solution is 0.70 mole
fraction in benzene and 0.30 mole fraction in toluene. The vapour pressure of
pure benzene and pure toluene are 75 mmHg and 22 mmHg respectively. Hence the
total vapour pressure is
Other Example of Ideal Solutions
(1)
Benzene + Toluene,
(2) n
hexane + n Heptane;
(3)
Chlorobenzene + Bromobenzene
(4) Ethyl bromide + Ethyl iodide;
(4)
n-Butyl chloride + n-Butyl bromide
(5) Ethyl alcohol + Methyl alcohol
(6) Tetrachloromethane + Tetrachlorosilane
REAL OR NON - IDEAL SOLUTIONS
Those solution which do not obey Raoult’s law
over entire range of composition and deviate from ideal behaviour, are real or
non – ideal solution.
Distinction between Ideal and Non Ideal Solutions:
Types of non-ideal solutions:
(1) Non
ideal solutions showing positive deviation
(2) Non ideal solutions showing negative
deviation.
(1) Non ideal solutions showing positive deviation:
(A)Characters for positive deviation:
When two liquids A and B on mixing form this
type of solution and show following characters
(1)
Non-ideal solution
showing positive deviation from Raoult’s law.
(2) A—B attractive force should be weaker
than A—A and B—B attractive forces.
(3) ‘A’ and ‘B’ have different shape,
size and character.
(4) ‘A’ and ‘B’ escape easily showing
higher vapour pressure than the expected value.
(5) The solution showing positive
deviations from ideal behaviour for those type of solutions,
(B) Condition of positive
deviation:
(C) Graph
of Positive deviation:
(D) Examples and cause of Positive
Deviation:
(A) Difference in
extent of association in two liquids
(1)
H2O and CH3OH (Methanol)
(2)
H2O and ROR’ (Ester)
(3)
H2O and CHCl3 (Chloroform)
Explanation:
Mixture of above pair produces a high distorted curve with maximum vapour pressure.
(B)
Association in one of the liquids through H-bonding
(4)
C2H5OH and C6H6
(Benzene)
(5)
ROH and ROR’ (Ester)
(6)
ROH and CHCl3 (Chloroform)
(7)
ROH CH3COCH3 (Acetone)
(8)
ROH and C6H12 (Cyclohexane)
Explanation:
(C)
Greater difference in length of hydrocarbon part of members of same homologous
series
(9)
n-butane and n-Heptane
(D)
Difference in polarity of
liquids: General Examples are when one is polar and other is non polar
(10)
CCl4 and CHCl3
Explanation:
(5)
Greater Difference in molar mass of non-polar molecules.
(11) CCl4
and C6H6
(13)
CCl4 and Toluene
(14)
Acetone and Benzene
(15)
CS2 and Acetone
(16)
CH3OH and Benzene
Explanation:
(2) Non ideal solutions showing negative deviation
(A) Characters for Negative deviation:
When two liquids
A and B on mixing form this type of solution and shows following character
(1) Non-ideal
solution showing negative deviation from Raoult’s law.
(2) A—B attractive force should be
greater than A—A and B—B attractive forces.
(3) ‘A’ and ‘B’ have different shape,
size and character
(4) Escaping tendency of both components
‘A’ and ‘B ’is lowered showing lower vapour pressure than expected ideally.
(B) Condition of Negative deviation:
The solution showing large negative deviations from ideal
behaviour and the vapour pressure of each component is considerably less than
that predicted by Raoult’s law, for these type of solutions.
(C) Graph
of Negative deviation:
(D) Examples and cause Positive
Deviation:
(1)
An acidic &
a basic liquid: Due to strong intermolecular hydrogen Bonding between
the proton of the acid & lone pair
of the donor atom of the basic liquid (C6H5OH & C6H5NH2)
(2)
Haloalkanes
(like chloroform) with more electronegative atoms: like oxygen or nitrogen or fluorine containing liquid (like ketones, esters, ethers, amines etc) due to
formation of Hydrogen – bonding between these.
Exception:
Excluding ALCOHOLS
which are highly associated and would show positive deviations.
(3)
Aqueous
solutions of strong volatile acids and water: For example sulfuric acid, nitric acid etc., which give non-volatile ions with
water
Newly form hydronium ions and Sulphate ions strongly
associated hence these solution show negative deviation
Subscribe to:
Comments (Atom)