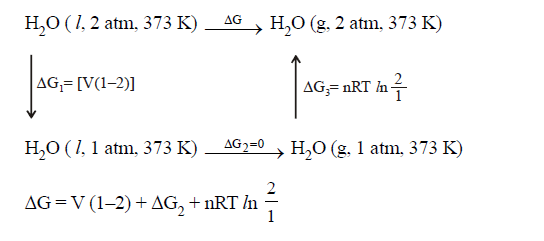Search This Blog
Friday, September 28, 2018
ENTHALPY (H) INTRODUCTION
We known 1st
law of thermodynamics
dU=dq+dW
If Pext=constant
Then dW= -PdV
and dU=dq- PdV
dq=dU-PdV
dq=(U2-U1)+P(V2-V1)
dq=(U2+PV2)-(U1+PV1)
dQ=H2-H1 ( H2=U2+PV2 and H1=U1+PV1)
dq=dH
hence
The enthalpy of a system is defined as:
H
= U + PV
DH = DU + D(PV)
Where
H is the enthalpy of the system
U is the internal energy of the system
P is the
pressure at the boundary of the system and its environment.
(1) In thermodynamics the quantity U + PV is a new state function and known
as the enthalpy of the system and is denoted by H=U+PV. It represents the total energy stored in the system.
(2) It may be noted that change in
enthalpy is equal to heat exchange at constant pressure.(3) Enthalpy
is also an extensive property as
well as a state function. (4)The absolute value of enthalpy cannot
be determined, however the change in enthalpy can be experimentally determined.
DH = DU + D(PV)
(5) Change in enthalpy is a more useful quantity than its
absolute value.
(6) The unit of measurement for enthalpy (SI) is joule.
(7)The enthalpy is the preferred expression of system energy
changes in many chemical and physical measurements, because it simplifies
certain descriptions of energy transfer. This is because a change in enthalpy
takes account of energy transferred to the environment through the expansion of
the system under study.
(8)The change dH
is positive in endothermic reactions,
and negative in exothermic
processes. dH of
a system is equal to the sum of non-mechanical work done on it and the heat
supplied to it.
(9) For quasi static processes under constant pressure, dH is equal to the change in the
internal energy of the system, plus the work that the system has done on its
surroundings. This means that the change in enthalpy under such conditions is
the heat absorbed (or released) by a chemical reaction.
NOTE:
Transfer of heat at constant volume brings about a change
in the internal energy(DU) of the system whereas that at constant pressure
brings about a change in the
enthalpy (DH) of the system.
For Ideal
gas
H=U+PV and U=f(T)
PV=nRT
H=U+nRT and H=f(T) only for ideal gas
For other
substance and real gas
H=U+PV
U=f(P,V,T)
and H=f(,PV,T)
So H=f(P,T)/ f(V,T)/ f(P,V)
H=f(T,P)
dH=(dH/dT)p dT+(dH/dP)T
dP------------------------------------- (1)
H=f(V,T)
dH=(dH/dV)T dV+(dH/dT)V
dT------------------------------------- (2)
H=f(P,V)
dH=(dH/dV)P dV+(dH/dP)V
dP------------------------------------- (3)
Out of the
above three relation H as function
on of (T,P) Has a greater significance. The above differential equation simplified
for different substance for different condition.
For
isobaric process : dP = 0
We known
QP=nCpmdT
(Molar Heat capacity at constant Pressure)
Cpm= (dQ/dT)P for 1 mole of gas
dQ=dH at dP=0
then Cpm= (dH/dT)P
For an ideal gas: change in
enthalpy at constant temperature with change in
pressure is zero. i.e.
Continue...........
THIRD LAW OF THERMODYNAMICS (TLOT)
We know that on increasing the entropy of a pure crystalline
substance increases because molecular motion increases with increase of
temperature and decreases on decreasing temperature. Or we can say that the entropy of a perfectly crystalline
solid approaches zero as the absolute zero of temperature is approached. Which
means that at absolute zero every crystalline solid is in a state of perfect
order and its entropy should be zero. This
is third law of thermodynamics.
We can calculate absolute value of entropy of any
substance in any state (solid ,liquid, gas) at any temperature by calculating
dS for the processes in going from the initial state to the state of the
substance for which entropy is to be calculated.
EXAMPLE: Find the entropy of change when 90 g
of H2O at 10C was converted into steam at 100C.(
Given Cp(H2O)=75.29 JK-1 and dHVap=43.932 k JK-1 mol-1)
SOLUTION:
EXAMPLE: Calculate dG for
(i) H2O
(l, 1 atm, 300 K ) ------à H2O (g, 1
atm, 300 K)
SOLUTION: (1)
(2)
WORK DONE (PV-WORK ANALYSIS )
Energy
that is transmitted from one system to another in such a way that difference of
temperature is not directly involved. This definition is consistent with our
understanding of work as dw= Fdx. The force (F) can arise from electrical, magnetic,
gravitational & other sources. It is a path function.
(1) Work done in Isothermal Irreversible Process:
(2) Work done in Isothermal Reversible Process:
(3) Work done in Adiabatic Irreversible Process:
(4) Work done in Adiabatic Reversible Process:
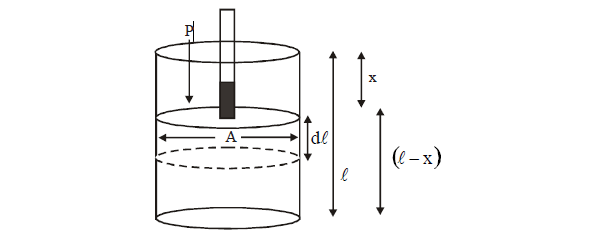
PV-Work analysis: Consider a cylinder fitted with a friction less piston, which enclosed no more of an
ideal gas. Let an external force F pushes the piston inside producing displacement in piston.
Let
distance of piston from a fixed point is x and distance of bottom of piston at
the same fixed point is
l. This
means the volume of cylinder = (l – x) A where A is area of cross section of
piston.
For a small displacement dx due to force F, work done on
the system.
dw = F.dx
Also F = PA
dW = PA.dx
V = (l – x)A
dV
= –A . dx
dW = –Pext. dV
Note :
(1): Litre atmosphere term is unit of energy. It is useful to remember the conversion:
1 litre atm= 101.3 Joules = 24.206 Cal.
(2): During expansion dV is positive and
hence sign of w is negative since work is done by the system and negative sign representing
decease in energy content of system. During compression, the sign of dV is negative
which gives positive value of w
representing the increase in energy content of system during compression.
EXAMPLE.1 mole of ideal monatomic gas at 27°C expands adiabatically against a constant external
pressure of 1.5 atm from a volume of 4dm3 to 16 dm3. Calculate
(i) q (ii) w and (iii) DU
SOLUTION: (i)
Since process is adiabatic \ q = 0
(ii) As
the gas expands against the constant external pressure.
W = - PVd=-P(V2-V1)
W =-1.5(16-4)
W= - 18 dm3
(iii) DU = q + w = 0 + (-18) = -18 atm dm3
(1) Work done in Isothermal Irreversible Process:
(2) Work done in Isothermal Reversible Process:
(3) Work done in Adiabatic Irreversible Process:
(4) Work done in Adiabatic Reversible Process:
Wednesday, September 26, 2018
SECOND LAW OF THERMODYNAMICS (SLOT)
(1) FLOT is law of conservation of energy, and according to it all chemical and physical processes take place in a such way that energy remain constant
(2) In FLOT we introduce enthalpy and internal energy.
(3)When two body at different temperatures are kept closed toeach other there will be transfer of heat while the transfer of heat take place called not explain by FLOT.
(4) we can not explain spontaneous and irreversible behaviour of processes by using FLOT.
In 2nd law of thermodynamics we will introduce entropy as criteria of spontaneity.
STATEMENT OF 2ND LAW :. "It is impossible to construct the heat engine which can take heat from a source and completely convert into work without creating any disturbance in the surrounding.
Subscribe to:
Posts (Atom)