Nucleophilic
substitution not occurs in aromatic compound but few Nucleophilic substitutions
occur in the presence of strong nucleophile. These reactions may be SN1
-Aromatic unimolecular and SN2 –Aromatic
bimolecular.
SN1 (Aromatic)
Type: The
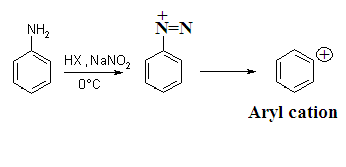
decomposition of diazonium salt in polar medium and formation of different
product in the presence of different nucleophile are example of Nucleophilic Aromatic
Substitution (SN1-Ar). The rate of reaction depends upon
only concentration of Aryl cation only hence
it is unimolecular reaction.
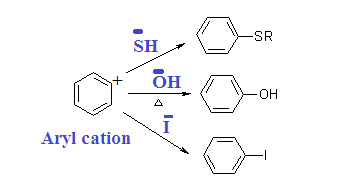
Aryl
cation is very reactive and takes up any nucleophile present recombined with
elimination of N2. Therefore reaction is reversible.
Although aryl cation is very unstable , the driving force for it formation of elimination of very stable due to very high bond dissociation of nitrogen molecule.

No comments:
Post a Comment