Search This Blog
Showing posts with label COORDINATION COMPOUNDS:. Show all posts
Showing posts with label COORDINATION COMPOUNDS:. Show all posts
Tuesday, June 23, 2020
What is denticity of NO and NO+ ligands ?
"NO" is a special ligand which can donate three electrons (3e) to the central metal atom in neutral state but in charge state (NO+) it can donate two electrons only to the central atoms.
Monday, June 15, 2020
What is stretching frequency of C-O bond in carbonyl Complexes?
V^co = 1/2πc√k/u
Where u= reduced mass of CO
k =force constant of CO bond
C= Velocity of light
V^CO = Stretching frequency of CO in carbonyl which increases energy to stretch CO along bond axis.
(1) Stretching frequency of CO is inversely proportional to negative oxidation state of central metal atom
(2) Extent of back bonding or back acceptance is directly proportional to negative oxidation state of central metal atom.
(3) M-C bond order is directly proportional to negative oxidation state of central metal atom
(4) C-O bond order is inversely proportional to negative oxidation state of central metal atom.
(5) d C-O bond length is directly proportional to negative oxidation state of central metal atom.
(6) Bond energy of C-O bond is inversely proportional to negative oxidation state of central metal atom
Sunday, February 2, 2020
What is Laporte Selection rule?
Laporte Selection Rule
is given by Otto Laporte a German American Physicist
Related Question:
According to Laporte
selection rule only allowed transitions are those occurring with a change in
parity (flip in the sign of one spatial coordinate.) OR During an electronic transition the azimuthal quantum number can
change only by ± 1 (Δ l = ±1) The Laporte selection
rule reflects the fact that for light to interact with a molecule and be
absorbed, there should be a change in dipole moment.
Practical meaning of the
Laporte rule:
Laporte allowed
transitions: are those which occur
between gerade to ungerade or ungerade to gerade orbitals.
Laporte forbidden transitions: are
those which occur between gerade to gerade or ungerade to ungerade orbitals.
Gerade = symmetric with respect to centre of inversion i.e.
atomic or molecular orbital with center of symmetry or number of nodal plane =
0, 2, 4 (even number)
Ungerade = anti symmetric with respect to centre of inversion
i.e. atomic or molecular orbital without center of symmetry or number of nodal
plane = 1, 3, 5, (odd numbers)
Important Note:
This rule affects
Octahedral and Square
planar
complexes as they have center of symmetry. Tetrahedral complexes do not have center of symmetry therefore this rule does not apply
Related Question:
Which of the
Complex of the following pairs has the highest value of CFSE?
Colour of
Complexes due to charge transfer:
Why violet
colour of [Ti(H2O)6]Cl3 disapear (colourless) on heating heating ?
Why [Ni(CN)4]-2
is colourless while [Ni(H2O)4]-2 although both have +2 oxidation state and 3d*8
configuration ?
Why [FeF6]3– is
colourless whereas [CoF6]3– is coloured ?
Why Fe(CO)5 is
colourless while Fe(bipy)(CO)3 is intensely purple in colour ?
Why all the
tetrahedral Complexes are high spin Complexes ?
Colour of Complexes due to charge transfer:
Why violet colour of [Ti(H2O)6]Cl3 disapear (colourless) on heating heating ?
Why [Ni(CN)4]-2 is colourless while [Ni(H2O)4]-2 although both have +2 oxidation state and 3d*8 configuration ?
Why [FeF6]3– is colourless whereas [CoF6]3– is coloured ?
Why Fe(CO)5 is colourless while Fe(bipy)(CO)3 is intensely purple in colour ?
Why all the tetrahedral Complexes are high spin Complexes ?
Although both [Mn(H2O)6]2+ and [FeF6]3- have a d5 configuration and high-spin complexes. But the dilute solutions of Mn2+ and Fe +3 complexes are therefore colorless. Why?
Both [Mn(H2O)6]2+
and [FeF6]3- have a d5 configuration
and high-spin complexes, but electronic transitions are not only
Laporte-forbidden, but also spin-forbidden. thus the dilute solutions of Mn2+
and Fe +3 complexes are colorless.
Important Note:
For
first transition series d5 system,
weak ligand field , and coordination number six (6) Complexes are found to be
colourless due to violation of selection rule.
What is the Selection rule for colour in complexes?
Spin
selection rule states that transitions that involve a change in spin
multiplicity as compare to ground state are forbidden.
Related Question:
(1) According to this rule,
any transition for which Δ S = 0 (it means no change in
spin multiplicity after d-d transition) is allowed.
(2) If Δ S ≠ 0 (change in spin multiplicity after transition) then it is forbidden
(transition not allowed)
Intensity of colour due to d-d
transition:
(1) Intensity of colour due
to d-d transition will found to be high if transition follow laporte selection
rule.
(2) Intensity of colour due
to d-d transition will found to be poor due violation of laporte selection
rule.
(3) Intensity of colour in
tetrahedral Complexes for (non centre of symmetry) is found to be higher than
octahedral (centre of symmetry).
Important Note:
For
first transition series d5 system, weak
ligand field, and coordination number six (6) Complexes are found to be
colourless due to violation of selection rule.
Related Question:
Which of the
Complex of the following pairs has the highest value of CFSE?
Colour of
Complexes due to charge transfer:
Why violet
colour of [Ti(H2O)6]Cl3 disapear (colourless) on heating heating ?
Why [Ni(CN)4]-2
is colourless while [Ni(H2O)4]-2 although both have +2 oxidation state and 3d*8
configuration ?
Why [FeF6]3– is
colourless whereas [CoF6]3– is coloured ?
Why Fe(CO)5 is
colourless while Fe(bipy)(CO)3 is intensely purple in colour ?
Why all the
tetrahedral Complexes are high spin Complexes ?
Colour of Complexes due to charge transfer:
Why violet colour of [Ti(H2O)6]Cl3 disapear (colourless) on heating heating ?
Why [Ni(CN)4]-2 is colourless while [Ni(H2O)4]-2 although both have +2 oxidation state and 3d*8 configuration ?
Why [FeF6]3– is colourless whereas [CoF6]3– is coloured ?
Why Fe(CO)5 is colourless while Fe(bipy)(CO)3 is intensely purple in colour ?
Why all the tetrahedral Complexes are high spin Complexes ?
Which of the Complex of the following pairs has the largest value of CFSE? (1) [Co(CN)6]3- and [Co(NH3)6]3+ (2) [Co(NH3)6]3+ and [CoF6]3- (3) [Co(H2O)6]3+ and [Rh(H2O)6]3+ (4) [Co(H2O)6]2+ and [Co(H2O)6]3+
(1) CN is the stronger
ligand than NH3 therefore CFSE of [Co(CN)6]3- will be more
than [Co(NH3)6]3+
Related Question:
(2) NH3 is stronger ligand than F therefore CFSE of [Co(NH3)6]3+ will be more than [CoF6]3-.
(3) Co belongs to 3d series whereas The Rh belong to 4d series. More the value of n more is CFSE therefore CFSE of [Rh(H2O)6]3+ is more than [Co(H2O)6]3+ .
(4) Oxidation number of Co in [Co(H2O)6]3+ is more than the Oxidation number of [Co(H2O)6]2+ therefore, CFSE of [Co(H2O)6]3+ is more than [Co(H2O)6]2+.
(3) Co belongs to 3d series whereas The Rh belong to 4d series. More the value of n more is CFSE therefore CFSE of [Rh(H2O)6]3+ is more than [Co(H2O)6]3+ .
(4) Oxidation number of Co in [Co(H2O)6]3+ is more than the Oxidation number of [Co(H2O)6]2+ therefore, CFSE of [Co(H2O)6]3+ is more than [Co(H2O)6]2+.
Related Question:
Which of the
Complex of the following pairs has the highest value of CFSE?
Colour of Complexes due to charge transfer:
Why violet colour of [Ti(H2O)6]Cl3 disapear (colourless) on heating heating ?
Why [Ni(CN)4]-2 is colourless while [Ni(H2O)4]-2 although both have +2 oxidation state and 3d*8 configuration ?
Why [FeF6]3– is colourless whereas [CoF6]3– is coloured ?
Why Fe(CO)5 is colourless while Fe(bipy)(CO)3 is intensely purple in colour ?
Why all the tetrahedral Complexes are high spin Complexes ?
Colour of Complexes due to charge transfer:
Why violet colour of [Ti(H2O)6]Cl3 disapear (colourless) on heating heating ?
Why [Ni(CN)4]-2 is colourless while [Ni(H2O)4]-2 although both have +2 oxidation state and 3d*8 configuration ?
Why [FeF6]3– is colourless whereas [CoF6]3– is coloured ?
Why Fe(CO)5 is colourless while Fe(bipy)(CO)3 is intensely purple in colour ?
Why all the tetrahedral Complexes are high spin Complexes ?
Sunday, January 19, 2020
Which of the Complex of the following pairs has the highest value of CFSE?
(1) [Co(CN)6]3- and [Co(NH3)6]3+
(1) Why all the tetrahedral Complexes are high spin Complexes?
(2) Why Fe(CO)5 is colourless while Fe(bipy)(CO)3 is intensely purple in colour ?
(3) Why [Mn(H2O)6]+2 is colourless although in which Mn+2 ion had five unpaired electrons ?
(4) Why [FeF6]3– is colourless whereas [CoF6]3– is coloured?
(5) Why [Ni(CN)4]-2 is colourless while [Ni(H2O)4]-2 is colour although both have +2 oxidation state and 3d8 configuration ?
(2) [Co(NH3)6]3+ and
[CoF6]3-
(3) [Co(H2O)6]3+ and [Rh(H2O)6]3+
(4) [Co(H2O)6]2+ and [Co(H2O)6]3+
(3) [Co(H2O)6]3+ and [Rh(H2O)6]3+
(4) [Co(H2O)6]2+ and [Co(H2O)6]3+
SOLUTION:
(1) CN is the stronger ligand than NH3 therefore CFSE of [Co(CN)6]3- will be more than [Co(NH3)6]3+
(2) NH3 is stronger ligand than F therefore CFSE of [Co(NH3)6]3+ will be more than [CoF6]3- .
(3) Co belong to 3d series whereas The Rh belong to 4d series. More the value of n more is CFSE therefore CFSE of [Rh(H2O)6]3+ is more than [Co(H2O)6]3+ .
(4) Oxidation number of Co in [Co(H2O)6]3+ is more than the Oxidation number of [Co(H2O)6]2+ therefore, CFSE of [Co(H2O)6]3+ is more than [Co(H2O)6]2+ .
(1) CN is the stronger ligand than NH3 therefore CFSE of [Co(CN)6]3- will be more than [Co(NH3)6]3+
(2) NH3 is stronger ligand than F therefore CFSE of [Co(NH3)6]3+ will be more than [CoF6]3- .
(3) Co belong to 3d series whereas The Rh belong to 4d series. More the value of n more is CFSE therefore CFSE of [Rh(H2O)6]3+ is more than [Co(H2O)6]3+ .
(4) Oxidation number of Co in [Co(H2O)6]3+ is more than the Oxidation number of [Co(H2O)6]2+ therefore, CFSE of [Co(H2O)6]3+ is more than [Co(H2O)6]2+ .
Related
Questions:
(1) Why all the tetrahedral Complexes are high spin Complexes?
(2) Why Fe(CO)5 is colourless while Fe(bipy)(CO)3 is intensely purple in colour ?
(3) Why [Mn(H2O)6]+2 is colourless although in which Mn+2 ion had five unpaired electrons ?
(4) Why [FeF6]3– is colourless whereas [CoF6]3– is coloured?
(5) Why [Ni(CN)4]-2 is colourless while [Ni(H2O)4]-2 is colour although both have +2 oxidation state and 3d8 configuration ?
Wednesday, August 28, 2019
Colour of Complexes due to charge transfer:
Colour originated by
charge transfer when electronic transition occurs from one part of the Complex
to other part i.e. Such type is also
called internal Redox reaction.
Intensity of colour in such type transition is very high as they do not require following any selection rule.
(A) Charge transfer from ligands to metal:
Examples , MnO4 - , MnO4 -2 , CrO4 -2, Cr2O7 -2 , [Fe(H2O)5(NO)]+2 , Na4[Fe(CN)5(NOS)].
Intensity of colour in such type transition is very high as they do not require following any selection rule.
(A) Charge transfer from ligands to metal:
Examples , MnO4 - , MnO4 -2 , CrO4 -2, Cr2O7 -2 , [Fe(H2O)5(NO)]+2 , Na4[Fe(CN)5(NOS)].
(B) Charge transfer from metal to ligands:
Examples, [Fe(CO)5] , [Fe (pi-C2H5)2], [Cr (pi-C6H6)2],
Examples, [Fe(CO)5] , [Fe (pi-C2H5)2], [Cr (pi-C6H6)2],
(C) Charge transfer from metal to metal:
Examples:
Examples:
(2) Prussian Blue:
Fe4[Fe(CN)6]3
IUPAC Name: Iron(III)hexacyanoferrate(II)
Common Name:
(Ferric ferrocyanide)
(2) Turnbull Blue: Fe3[Fe(CN)6]2
IUPAC Name: Iron(II)hexacyanoferrate(III)
Common Name:
(Ferrous ferricyanide)
Related Question:
Topic:
COORDINATION COMPOUNDS:
Why violet colour of [Ti(H2O)6]Cl3 disapear (colourless) on heating heating ?
[Ti(H2O)6]Cl3 is an octahedral violet colour complex; the violet colour of this
complex is due to d-d transition of a single (d1) electron from t2g lower level to eg higher
level by absorbing corresponding energy of yellow green region of light and
emitted energy corresponding to violet region and hence appears violet in
colour. But on heating it is dehydrated and water molecules (ligand)
removed so in absence of ligand splitting of D orbitals does not occur hence it
becomes colourless.
Similarly anhydrous CuSO4 is white ,
but CuSO4 .5H2O is Blue in Colour.
Related Question:
Related Question:
Which of the
Complex of the following pairs has the highest value of CFSE?
Colour of Complexes due to charge transfer:
Why violet colour of [Ti(H2O)6]Cl3 disapear (colourless) on heating heating ?
Why [Ni(CN)4]-2 is colourless while [Ni(H2O)4]-2 although both have +2 oxidation state and 3d*8 configuration ?
Why [FeF6]3– is colourless whereas [CoF6]3– is coloured ?
Why Fe(CO)5 is colourless while Fe(bipy)(CO)3 is intensely purple in colour ?
Why all the tetrahedral Complexes are high spin Complexes ?
Colour of Complexes due to charge transfer:
Why violet colour of [Ti(H2O)6]Cl3 disapear (colourless) on heating heating ?
Why [Ni(CN)4]-2 is colourless while [Ni(H2O)4]-2 although both have +2 oxidation state and 3d*8 configuration ?
Why [FeF6]3– is colourless whereas [CoF6]3– is coloured ?
Why Fe(CO)5 is colourless while Fe(bipy)(CO)3 is intensely purple in colour ?
Why all the tetrahedral Complexes are high spin Complexes ?
Topic:
COORDINATION COMPOUNDS:
COLOURS OF COMPLEXES AND SELECTION RULE:
Electronic
transitions in a complex are governed by Selection rules A selection rule is a
quantum mechanical rule that describes the types of quantum mechanical transitions
that are permitted.they reflect the restrictions imposed on the state changes
for an atom or molecule during an electronic transition. Transitions not
permitted by selection rules are said forbidden, which means that theoretically
they must not occur (but in practice may occur with very low probabilities).
Related Question:
(1) Laporte Selection Rule:
Laporte
Selection Rule is given by Otto Laporte a German American Physicist
According to
Laporte selection rule only allowed transitions are those occurring with a change
in parity (flip in the sign of one spatial coordinate.) OR During an electronic transition the azimuthal
quantum number can change only by ± 1 (Δ l = ±1) The Laporte
selection rule reflects the fact that for light to interact with a molecule and
be absorbed, there should be a change in dipole moment.
Practical meaning of the
Laporte rule:
Laporte allowed transitions: are those which occur between gerade to ungerade or ungerade to gerade orbitals.
Laporte forbidden transitions:
are those which
occur between gerade to gerade or ungerade to ungerade orbitals.
Gerade = symmetric with respect to centre of
inversion i.e. atomic or molecular orbital with center of symmetry or number of
nodal plane = 0, 2, 4 (even number)
Ungerade = anti symmetric with respect to centre
of inversion i.e. atomic or molecular orbital without center of symmetry or number
of nodal plane = 1, 3, 5, (odd numbers)
Important Note:
This rule affects Octahedral and Square planar
complexes as they have center of symmetry. Tetrahedral
complexes do not have center of symmetry therefore
this rule does not apply
(2) Spin Selection Rule:
Spin selection
rule states that transitions that involve a change in spin multiplicity as compare to ground state are forbidden.
(1) According to this rule, any transition for which Δ S = 0 (it means no change in spin multiplicity after d-d transition) is allowed.
(2) If Δ S ≠ 0 ( change in spin multiplicity after transition) then it is forbidden (transition not allowed)
ILLUSTRATIVE
EXAMPLE: [Mn(H2O)6]2+
and [FeF6]3- both have a d5 configuration
and high-spin complexes. Electronic transitions are not only Laporte-forbidden,
but also spin-forbidden. The dilute solutions of Mn2+ and Fe +3
complexes are therefore colorless
Important Note:
For
first transition series d5 system, weak
ligand field, and coordination number six (6) Complexes are found to be
colourless due to violation of selection rule.
Related Question:
Which of the
Complex of the following pairs has the highest value of CFSE?
Colour of
Complexes due to charge transfer:
Why violet
colour of [Ti(H2O)6]Cl3 disapear (colourless) on heating heating ?
Why [Ni(CN)4]-2
is colourless while [Ni(H2O)4]-2 although both have +2 oxidation state and 3d*8
configuration ?
Why [FeF6]3– is
colourless whereas [CoF6]3– is coloured ?
Why Fe(CO)5 is
colourless while Fe(bipy)(CO)3 is intensely purple in colour ?
Why all the
tetrahedral Complexes are high spin Complexes ?
Colour of Complexes due to charge transfer:
Why violet colour of [Ti(H2O)6]Cl3 disapear (colourless) on heating heating ?
Why [Ni(CN)4]-2 is colourless while [Ni(H2O)4]-2 although both have +2 oxidation state and 3d*8 configuration ?
Why [FeF6]3– is colourless whereas [CoF6]3– is coloured ?
Why Fe(CO)5 is colourless while Fe(bipy)(CO)3 is intensely purple in colour ?
Why all the tetrahedral Complexes are high spin Complexes ?
Topic:
COORDINATION COMPOUNDS:
Why [Ni(CN)4]-2 is colourless while [Ni(H2O)4]-2 although both have +2 oxidation state and 3d*8 configuration ?
In case of [Ni(CN)4]-2 Ni is in +2 and
3d8 configuration but presence
of strong ligand (CN) , the two unpaired electron in the 3d orbital pair up
thus there is no unpaired electrons present hence it is colourless.
In case of [Ni(H2O)4]+2 Ni is in +2 and
3d8 configuration and two
unpaired electrons which I do not pair up in the presence of weak ligand
(H2O) , hence it is colour due to d-d transition , red colour light is absorbed and give it
complementary colour green.
Topic:
COORDINATION COMPOUNDS:
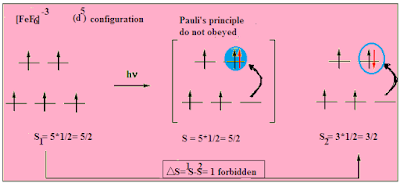
Why [FeF6]3– is colourless whereas [CoF6]3– is coloured ?
[FeF6]3- is a Fe (III) complex
hence [Ar] 3d5. F-
is a weak field ligand and the complex is high spin:
t2g (↑) (↑)(↑) →Δoct→ (↑)(↑)eg.
And there is 5
unpaired electrons hence it's spin multiplicity = (2S+1) = 6 , and multiplicity of a excited is cannot be six thus The transitions in Fe +3 ion are spin
forbidden and are extremely weak so as to make [FeF6]3-
almost colorless.
[CoF6] 3- this is a Co (III)
complex [Ar] 3d6. Again F-
is a weak field ligand and [CoF6]3- is one of only two
common cmplxs that is high spin:
t2g (↑↓) (↑)(↑) →Δoct→ (↑)(↑) eg
Only one band is
expected namely the t2g→eg transition (d1). The complex
is blue consistent with this
transition being in the low energy red.
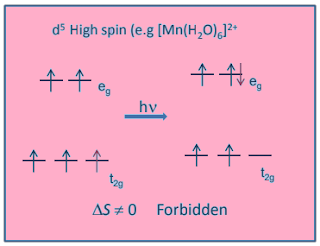
Why [Mn(H2O)6]+2 is colourless although in which Mn+2 ion had five unpaired electrons ?
There are 5
unpaired electrons in Mn+2 ion and we know that.
Spin multiplicity is = (2S+1)
Where S is some of spin thus S = 5×1/2=5/2
Hence multiplicity = (2S+1)= 6
Spin multiplicity is = (2S+1)
Where S is some of spin thus S = 5×1/2=5/2
Hence multiplicity = (2S+1)= 6
we know multiplicity of excited state cannot be six, thus electronic transition in Mn+2 are spin forbidden, hence [Mn(H2O)6]+2 salt appear colourless.
Also [Mn(H2O)6]+2
has centre of symmetry and
in such cases electronic transition are expected to be laporate forbidden.
Topic:
COORDINATION COMPOUNDS:
Subscribe to:
Posts (Atom)