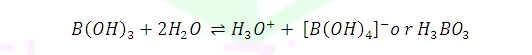H3BO3 is soluble in water and behaves as weak monobasic acid. It does not donate protons but rather it accepts OH- .Therefore it acts as a Lewis acid [B(OH)3] .It is not a proton donor because it accept lone pair or hydroxyl ion from water.
Related Questions:
Why Ga has small size than Al exceptionally
Why aqueous solution of borax reacts with two moles of acids ?
What is structure of solid Ortho Boric acid ?
What is the structure of trimetaboric acid and trimetaborate ion?
Why Borazine is more reactive than benzene towards Electrophic Aromatic substitution reactions ?
Why Borazine (B3N3H6) is also known as inorganic benzene ?.
Why B-F bond length in BF3 is shorter (130 pm) than B-F bond Iength in BF4- (143 pm)?. Explain.
Why B-F do not exist as dimer?. Explain.
Although anhydrous aluminium chloride is covalent but its aqueous solution is ionic in nature. Why?
Why Boric acid become strong acid in the presence of cis 1,2-diol or 1,3-diol ?