Pyridine is more basic as compared to pyridazine,
pyrimidine and pyrazine because pyridazine, pyrimidine and pyrazine containing
two nitrogen atoms which are exerted inductive effect to each other so
availability of lone pair for donation is less.
But
pyridazineis more basic as expected, it may be due to
electrostatic repulsion of the lone pairs of the vicinal nitrogens. This
repulsion destabilizes the non-protonated structure and therefore makes
protonation easier.
Related Questions:
(1) Give an
explanation for the fact that Guanidine NH=C(CH3)2 is a stronger base than most
of amines?
(2) Arrange in
correct order of basic Character of aniline, pyrrol, pyridine and piperidine?
(3) What is correct
basicity order of pyridine, pyridazine, pyrimidine and pyrazine ?
(4) Why pyridine is
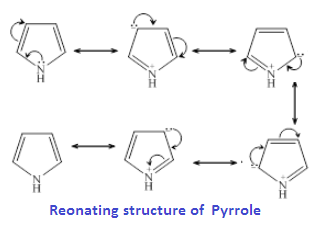
more basic than Pyrrole?
(5) Why pyrimidine
is less basic than pyridine?
(6) Imidazole is
more basic than pyridine? Why?
(7) Biological
Important of Imidazole and structure:
(8) Pyridine is
almost 1 million times less basic than piperidine? Why?
(9) Cyclohexylamine
amine is the stronger base than Aniline? Why?
(10) Tetrahydroquinoline
amine is the stronger base than Tetrahydroisoquinoline? Why?