(1) SN-Ar-(Substitution of hydrogen of benzene):
(3) Substitution of unactivated hydrogen: Benzyne
Intermediate Mechanism:
SN-Ar-Elimination-Addition: Benzyne Intermediate Mechanism:
An
aryl halide can undergo a nucleophilic substitutoin reaction in the presence of
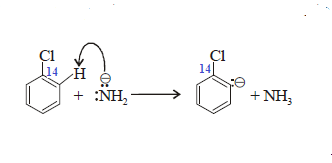
a very strongbase such as NH2 When chlorobenzene – that has the carbon to which chlorine is
attached isotopically labeled with Cabon-14 –is treated with amide
ion in liquid ammonia, aniline is
obtained as a product. Half of the product has the amino group attached to the
isotopically labelled carbon (14) as expected, but the other half has the amino
group attached to the carbon adjacent to the labelled carbon.
The mechanisms that accounts for the
experimental observations involves formation of a benzene intermediate which has
two equivalent carbon atoms to which amino group can be attached. Benzyne has
an extra (Pi) bond between two adjacent carbon atoms of benzene and can be
formed as
Step-(1): Strong base NH2- removes a proton
from the position ortho to halogen:
Step-(2): Anion formed in step (1) eliminates the halide
ion, thereby forming Benzyne:
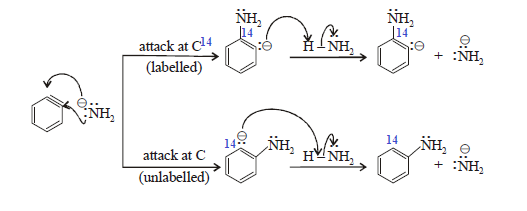
The incoming nucleophile can attack
either of the carbons of the “triple bond” of benzyne. Protonation of the
resulting anion form the substitution product. The overall reaction is an
elimination-addition reaction; benzyne is formed in an elimination reaction and
immediately undergoes an addition reaction.
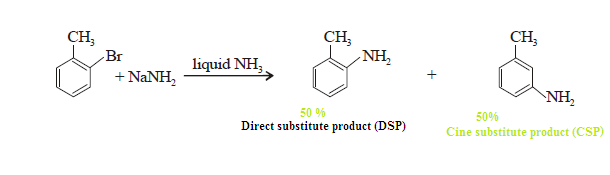
Substitution at the carbon (C-14) that
was attached to the leaving group is called direct substitution product (DSP).
Substitution at the adjacent labeled carbon (C-14) of is called cine
substitution product (CSP).
Characteristic of Benzyne reaction:
(1) It is a SN-EA reaction proceed via elimination –Addition Nucleophilic Substitution reaction.
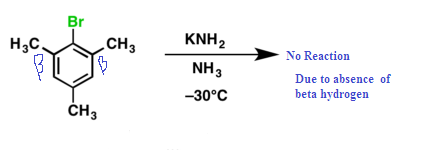
(2) Aryl halide + strong base required
(3) Presence of beta hydrogen must be needed.
(4) NaNH2 and KNH2 can be considered to be
essentially the same for our purposes
(5) For symmetrical intermediate which
is attacked equally on either side. so that the roughly 50:50
ratio of products are obtained .



No comments:
Post a Comment